Overview
This article discusses approaches to the power architecture used in medical devices that connect to patients. Key factors to consider include insulation, leakage/contact current, electromagnetic compatibility (EMC), the number of required power outputs, and the intended operating environment.
Application parts
Components that contact the patient are referred to as application parts. An application part is a portion of the medical device that directly contacts the patient or may contact the patient during normal use in order for the device to perform its function.
Classification and standards
The 60601-1 safety standard defines application parts based on the type of patient contact and the nature of the medical device. The current edition of 60601-1 was first published in December 2005 (3rd edition) and has been adopted in major regions as follows:
- IEC 60601-1:2005 (3rd edition + CORR. 1:2006 + CORR. 2:2007 + A1:2012)
- Europe: EN 60601-1:2006/A1:2013/A12:2014
- United States: ANSI/AAMI ES60601-1: A1:2012, C1:2009/(R)2012 and A2:2010/(R)2012
- Canada: CSA CAN/CSA-C22.2 No. 60601-1:14
Each classification in the standard includes requirements for protection against electric shock. The three classifications, listed from least to most stringent, are:
- B (Body). For application parts that are generally non-conducting and may be earthed.
- BF (Body Floating). For application parts that are electrically connected to the patient and must be floating and not earthed; excludes parts with direct cardiac contact.
- CF (Cardiac Floating). For application parts that connect directly to the heart. CF parts must be floating and not earthed.
Insulation requirements
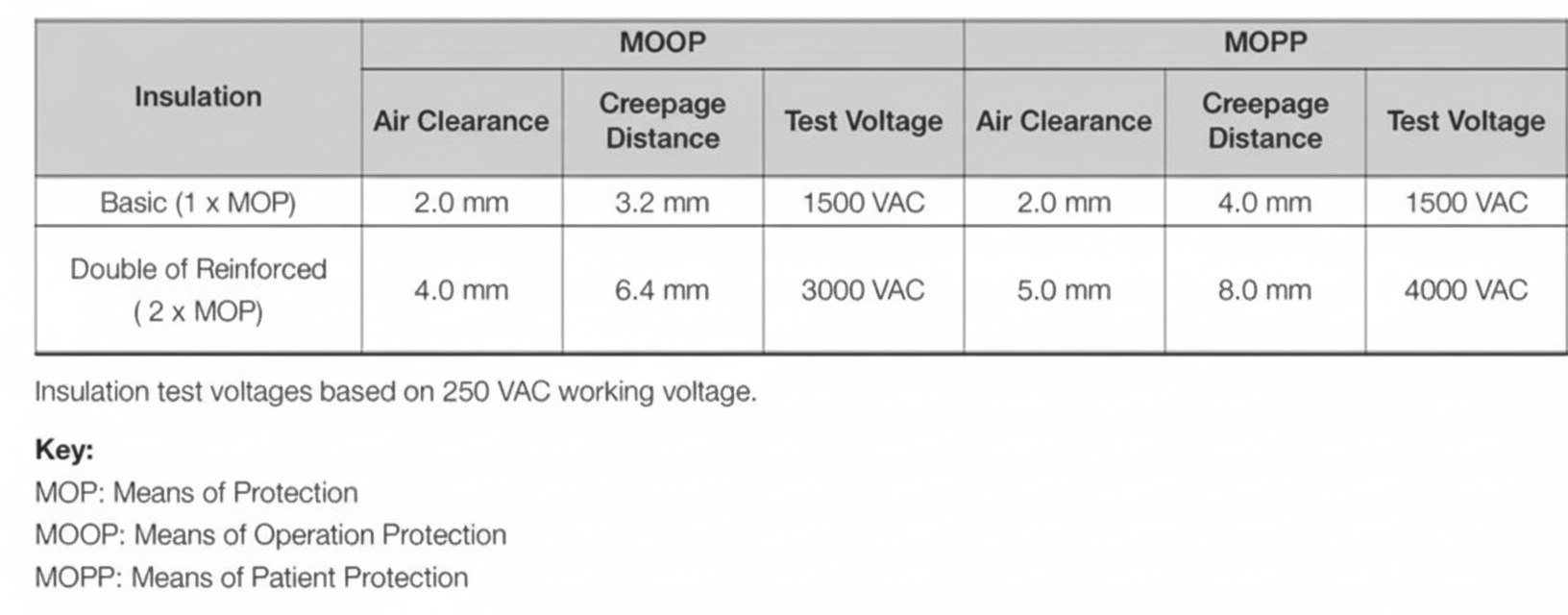
Patient-connected medical devices must provide means of protection (MOP) to prevent application parts and other accessible parts from exceeding voltage, current, or energy limits. A protective earth connection counts as one MOP, basic insulation provides one MOP, and reinforced insulation provides two MOP. Protection can be classified as means of operator protection (MOOP) or means of patient protection (MOPP). Patient-connected equipment requires 2 × MOPP.
For BF and CF devices, the power architecture must provide one or two stages delivering 2 × MOPP, one stage to earth delivering 1 × MOPP, plus additional safety insulation from a secondary output to earth rated as 1 × MOPP, all referenced to the (maximum rated) mains input voltage.
Leakage current
Power systems must be designed to limit touch current, patient auxiliary current, and patient leakage current. The maximum allowable touch current is 100 μA under normal conditions and 500 μA under a single fault condition. This effectively limits chassis leakage current during normal operation to 500 μA. Maximum allowable patient auxiliary current and patient leakage current are specified in the relevant tables of the standard.
When a patient-connected device requires electrical connections, the designer of the power architecture must ensure the power system provides the required safety insulation while minimizing leakage current by isolating the patient from earth during normal operation and fault conditions, and provide appropriate protective measures.
Electromagnetic compatibility (EMC)
Medical devices must also meet EMC requirements described in IEC 60601-1-2. The latest edition, published in 2014 and commonly referred to as Edition 4, has two main objectives.
First, it increases immunity requirements to account for the prevalence of wireless communication devices operating near critical medical equipment. These devices may include mobile phones, Bluetooth, WiFi, DECT, RFID, or paging systems.
Second, it introduces a risk analysis element to determine the appropriate level of immunity based on the device, its intended operating environment, and foreseeable interference. The standard now addresses environments beyond professional healthcare facilities, including home healthcare and special environments where high-power fields may be generated. Because immunity requirements are linked to environment rather than to specific product labels such as "life-support," manufacturers must identify intended use and select appropriate immunity levels to mitigate risk of malfunction.
Power schemes for patient-connected devices
In BF and CF class devices, the power system is a critical element for meeting insulation, leakage current, and EMC requirements.
For home healthcare environments, a practical non-earthed insulation approach is to implement a naturally floating Class II insulation system. It remains important to meet chassis and patient-to-earth leakage requirements. This approach is practical for products up to roughly 300 W; above 300 W, EMC requirements become harder to manage.
Most standard AC-DC power supplies that comply with medical safety approvals are not suitable for direct patient connection for several reasons:
- They lack the required output-to-earth insulation.
- They do not meet patient leakage current limits.
- Although they may provide the required 2 × MOPP from input to output and 1 × MOPP from input to earth, many supplies use operational insulation from output to earth rated near 500 VAC/VDC with a required test voltage of 1500 VAC; therefore, creepage and clearance distances must be observed when basic insulation at mains voltage is needed for patient-connected applications.
- Input-to-output parasitic capacitance can be too high, leading to excessive output-to-earth leakage current.
Secondary-insulated DC-DC solution
For low-power patient-connected devices, a simple and low-cost solution is to use a secondary-insulated DC-DC converter that is medically recognized. Such converters provide basic insulation at mains voltage on their inputs and minimize input-to-output capacitance (typically 20–50 pF), reducing potential patient leakage currents to single-digit microamps. This approach also addresses possible low-integrity system I/O signals that may connect to uncontrolled external equipment such as computers or monitors.
In a simplified model of the power system, the patient leakage path shows capacitors C4 and C5 in series. C5 represents the DC-DC converter's input-to-output capacitance, which is very small and presents high impedance to reduce leakage; C4 is typically larger.
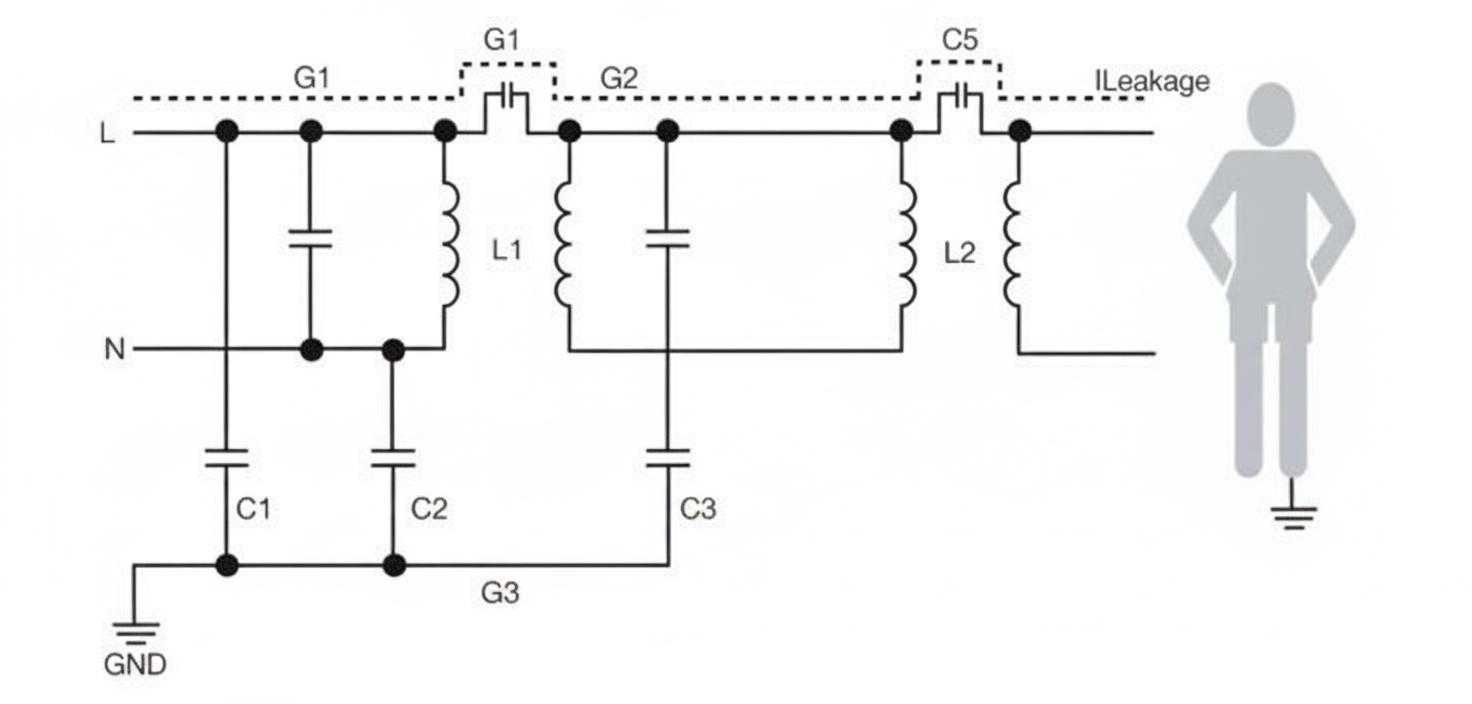
Figure 1: Simplified model of a power system with secondary DC-DC insulation
Medically approved DC-DC converters rated from 1 W to 20 W are commercially available with the required input-to-output insulation and very low internal capacitance. When used with a medically approved AC-DC input supply, patient leakage current can be reduced to below 2 μA, suitable for BF and CF applications. For required power below about 2–3 W, fixed-input, unregulated DC-DC converters fed from a regulated AC-DC front end can provide a cost-effective solution.
There are DC-DC products available that provide tightly regulated outputs across wide DC input and load ranges, with 2 × MOPP insulation and similarly low internal capacitance, making them suitable for battery-powered or DC-input portable devices.
Multi-output power schemes
For devices that require multiple outputs for patient-connected circuits, adding secondary DC-DC converters can provide very low leakage currents (on the order of 2 μA) and offer a robust path to meeting patient auxiliary and leakage current requirements.
If the AC-DC stage requires multiple outputs, chassis leakage can become constrained by the single fault contact current limits, which may render multiple isolated supplies impractical. In that case, a multi-output AC-DC supply may be required.
For low-power systems in the 2–300 W range, there are various medical-approved bare-board or U-channel output modules available. Alternatively, additional voltage rails can be created using isolated or non-isolated DC-DC converters fed from a single-output AC-DC supply.
For high-power applications, modular configured solutions are available that provide high-power, multi-output capabilities and meet medical safety approvals.
Motor-driven and high-power applications
In high-power and motor-driven applications such as bone saws, surgical tools, and powered tables, beds, or chairs, it can be impractical to add an additional isolation stage due to limited availability of high-power DC-DC devices with the required insulation and because of the efficiency penalty of double conversion. These applications require a power design that provides the necessary insulation, spacing, and patient leakage performance.
The combination of high insulation and low leakage current presents design challenges for AC-DC supplies. Increased clearance and creepage on the secondary side are required, and these must be addressed in the context of system integration. Low emissions and low leakage requirements can conflict: low-noise topologies and careful management of differential and common-mode noise are needed, while minimizing low-frequency perturbations in the primary circuit reduces patient leakage via input-to-output capacitances.
These high-power applications are commonly classified as BF rather than CF, so the leakage current requirement is less stringent (for example, 100 μA rather than 10 μA). Increasingly, standard AC-DC supplies are available that can be adapted to BF applications. For example, a 250 W BF-class AC-DC supply may meet insulation, leakage current, and Edition 4 EMC requirements while providing convection-cooled operation to avoid system fans and combining constant-current overload behavior with short-duration peak-power capability, making it suitable for motor-driven applications.

Figure 2: Example 250 W convection-cooled AC-DC supply suitable for BF applications
Design considerations and conclusion
Designing power systems for patient-connected medical devices is challenging. Using standardized, approved, and appropriately rated components or verified combinations that address EMC performance can simplify compliance and reduce risk. Such an approach supports safer device integration and faster time to market while meeting insulation, leakage current, and EMC requirements.