Introduction
On January 3, 2019, the Chang'e 4 lunar probe achieved the first soft landing on the far side of the Moon. The landing site was a rugged polar region, where sunlight is more limited and nighttime temperatures are lower. To avoid mission failure due to freezing, the probe carried, in addition to solar panels, a radioisotope battery to enable scientific observations during lunar night.
Radioisotope Batteries vs. Nuclear Power Plants
When people hear "nuclear power," they often think of nuclear power plants. The only commonality between a nuclear power plant and a radioisotope battery is the use of nuclear technology; their operating principles are fundamentally different. A nuclear power plant uses heat from nuclear fission to generate steam that drives turbines, similar to fossil-fuel power plants except the heat source differs. A radioisotope battery uses heat from the natural decay of radioisotopes and converts it to electricity via thermoelectric effects or other energy conversion mechanisms.
Definition and Classification
Radioisotope batteries, also called isotope batteries, convert the energy released by radioactive decay particles (such as alpha particles, beta particles, and gamma rays) into electrical energy.
By output voltage, they can be divided into high-voltage types (hundreds to thousands of volts) and low-voltage types (tens of millivolts to around 1 V). By energy conversion mechanism, they are classified as direct-conversion and indirect-conversion devices. Specific types include direct charge nuclear batteries, gas ionization batteries, radiation-voltaic effect batteries, phosphor-photovoltaic batteries, thermally induced photovoltaic batteries, thermoelectric batteries, thermionic emission batteries, electromagnetic radiation energy conversion batteries, and heat-engine conversion batteries. Direct-charge and gas-ionization batteries are direct-conversion types and are less commonly used. The most widely applied types today are thermoelectric (thermocouple) radioisotope batteries and heat-engine conversion batteries.
Historical Progress
Substantial progress on radioisotope batteries began in the 1950s. Their small size, light weight, and long service life, along with output power that is insensitive to external temperature, chemical environment, pressure, or electromagnetic fields, make them suitable for operation over wide temperature ranges and in harsh environments. Radioisotope batteries have been applied in aerospace, polar stations, and medical implants such as pacemakers.
Basic Working Principle
The heat source of a radioisotope battery is a radioactive isotope. During decay, isotopes release energy in the form of radiation that has much greater energy than ordinary materials. Two key attributes of this energy are: 1) the decay energy and decay rate are unaffected by external temperature, chemical reactions, pressure, or electromagnetic fields, resulting in high interference resistance and reliable operation; and 2) the long decay time of many isotopes yields long battery lifetimes.
Common isotopes used include strontium-90 (Sr-90, half-life 28 years), plutonium-238 (Pu-238, half-life about 89.6 years), and polonium-210 (Po-210, half-life about 138.4 days). In a typical cylindrical battery, the fuel is placed at the center and surrounded by thermoelectric elements. The isotope emits high-energy alpha particles that heat the thermoelectric elements, which convert heat into electric current.
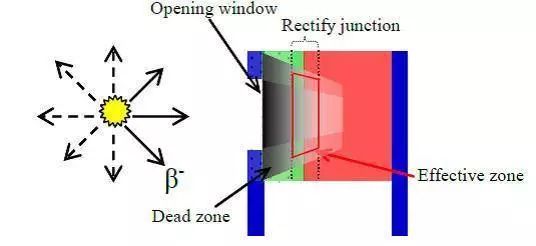
The core of an isotope battery is an energy transducer. A commonly used transducer is a static thermoelectric converter that relies on thermocouple principles to produce a voltage between different metals, thereby generating electricity. Thermoelectric isotope batteries can be made very small but have low conversion efficiency, typically 10% to 20%, with the majority of heat lost. Typical battery structure from outside in includes an outer alloy shell for protection and heat dissipation, a radiation shielding layer to prevent leakage, a transducer layer where heat is converted to electricity, and a core where the radioactive isotope continually decays and emits heat.
Energy Conversion Mechanisms and Voltage Classes
By conversion mechanism, radioisotope batteries are split into direct-conversion and indirect-conversion types. Specific mechanisms include direct charge, gas ionization, radiation-voltaic effect, phosphor-photovoltaic, thermally induced photovoltaic, thermoelectric, thermionic emission, electromagnetic radiation energy conversion, and heat-engine conversion.
By output voltage, they fall into high-voltage types (hundreds to thousands of volts) and low-voltage types (tens of millivolts to about 1 V). High-voltage batteries often use beta-emitting sources (such as Sr-90 or tritium) as emitters and a thin carbon-coated nickel collector as the counterelectrode, with a vacuum or solid dielectric between them. Low-voltage types are subdivided into thermoelectric stacks, gas ionization structures, and phosphor-photovoltaic structures.
Major Development Milestones
The first radioactive isotope battery was built on January 16, 1959. It weighed 1.8 kg and produced about 11.6 kWh over 280 days. After that, development accelerated. In 1961, the first U.S. satellite linked to radioisotope power used an isotope battery for its radio transmitter. In 1976, the U.S. Viking landers on Mars used radioisotope power systems. In 2012, the Curiosity rover, weighing over 900 kg, was powered by a multi-mission radioisotope thermoelectric generator (RTG) that contains a Pu-238 dioxide heat source and solid-state thermoelectric elements; the system was designed for a 14-year service life and exceeds typical solar-panel performance under Martian conditions.
Materials Used in Radioisotope Batteries
Typical battery structure places the radioactive isotope at the core, surrounded by energy conversion materials, then radiation shielding, and finally an outer alloy shell for protection and heat dissipation. Materials therefore cover isotope fuel, conversion materials, radiation shielding, and heat-management components. The special mission requirements dictate the use of specialized materials.
Isotope Fuel Sources
The role of the isotope source varies by battery type. Direct-charge batteries use emitted charged particles to create a potential difference. Gas-ionization and radiation-voltaic batteries use ionization effects on the medium. Phosphor-photovoltaic batteries use emitted radiation to excite phosphors, whose light is converted to electricity. Thermally driven photovoltaic, thermoelectric, and heat-engine batteries use decay heat for energy conversion.
Isotope sources used as energy carriers must meet criteria: long half-life for longevity, high power density, low radiological hazard, ease of fabrication, cost-effectiveness, and ease of shielding. Based on emitted radiation, isotopes are grouped as alpha, beta, or gamma sources. About ten isotopes are commonly suitable, including gamma emitter Co-60; beta emitters Sr-90, Cs-137, Ce-144, and Pm-147; and alpha emitters Po-210, Pu-238, Am-241, Cm-242, and Cm-244.
These isotopes are typically sealed in high-temperature-resistant shells to form the energy core. For space applications, alpha heat sources such as Pu-238 and Po-210 are preferred due to low external radiation dose and minimal shielding mass requirements. Pu-238 has a half-life around 87.7 years and releases about 5.48 MeV per decay. The United States has used Pu-238 sources in spaceflight. Fuel forms evolved from metallic plutonium to plutonium oxide microspheres, oxide-molybdenum ceramics, and now hot-pressed Pu-238O2 blocks for general-purpose RTGs.
Energy Conversion Materials
Different battery types use different conversion materials. In direct-charge batteries, both electrodes are metals: the emitter that emits electrons becomes the positive electrode, and the collector becomes the negative electrode. Researchers have studied combinations such as copper plates paired with Ni-63 isotope plates; Ni-63 emits beta particles and, upon losing electrons, becomes positively charged while the copper plate receives electrons, producing a current when connected to a load. Ni-63 has a half-life of about 100 years, implying long operational life.
Gas-ionization batteries rely on materials with different work functions. High work-function materials include platinum, lead oxide, molybdenum, and gold; low work-function materials include magnesium and aluminum. Radioactive gas dielectrics include tritium or Kr-85. Using lead dioxide (high work function) and magnesium (low work function) electrodes can yield open-circuit voltages around 1.5 V.
Advances in radiation-voltaic, phosphor-photovoltaic, thermally induced photovoltaic, and thermoelectric batteries are closely linked to semiconductor technology improvements. For example, the U.S. Department of Energy's Advanced Radioisotope Power Systems program explored thermally induced photovoltaic converters using GaSb, with Ge and GaAs also meeting requirements. Such semiconductor-based converters can have 2–3 times the efficiency of current thermoelectric systems, reducing required isotope mass, battery weight, and cost for the same output power.
Thermoelectric radioisotope batteries use thermoelectric materials as the conversion medium, which is crucial to converting decay heat into electricity. Common thermoelectric semiconductors include bismuth telluride, lead telluride, germanium-silicon alloys, and chalcogenide compounds. P-type and N-type semiconductor elements are connected in series to form a thermoelectric stack. For example, sealing about 11 mg of Po-210 in a 10 mm diameter sphere combined with seven chrome-nickel-constantan thermocouples can produce a temperature difference of 78°C and an open-circuit voltage of 42 mV. Historically, PbTe was used; later TAGS (Te-Ag-Ge-Si) alloys were developed for P-type legs with PbTe as N-type, providing hot-junction temperatures around 500–610°C. More recently, SiGe has been used in 100 W-class and general-purpose thermoelectric batteries, enabling hot-junction temperatures up to 1000°C.
Sealing and Protective Materials
Because radioisotope batteries often operate in harsh conditions and must be fail-safe to prevent leakage and radiological incidents, sealing and protection include fuel particle cladding, radiation shielding outside the conversion layer, and outer casings. Typical sealing materials are metal alloys, carbon-based materials, and ceramics.
Pu-238O2 fuel particles used in space are clad with thin pyrolytic carbon layers (~5 μm) and zirconium carbide (ZrC) layers (>10 μm). These coated particles are dispersed in a graphite matrix and pressed into fuel pellets. The graphite matrix provides good thermal conductivity and buffers stress; the porous pyrolytic carbon stores helium generated by Pu decay and absorbs stress; the ZrC layer resists high temperatures and contains released helium, preventing fuel leakage and improving space battery safety.
Designs for thermoelectric radioisotope batteries have used pyrolytic graphite and ZrC-clad PuO2 fuel pellets pressed into compacts wrapped in Pt-30%Rh foil. The outer region uses pyrolytic graphite as thermal insulation and a carbon-carbon composite buffer layer as the outermost protective layer. The outer case is often aluminum alloy, with multilayer insulation and thermoelectric conversion materials between the case and the heat source.
In a thermophotovoltaic radioisotope design, each general-purpose heat source module may include multiple 62.5 W PuO2 fuel assemblies sealed in an iridium alloy capsule. Graphite modules protect the iridium liner during prelaunch and accident scenarios. These layers typically include dense carbon layers and a buffer carbon layer made from finely woven, high-temperature carbon-carbon composite. Dense layers prevent liner fracture on landing; the buffer layer provides ablation protection during inadvertent reentry. High-temperature insulation between dense and buffer layers uses carbon fiber reinforced carbon composites to prevent thermal shock and airflow-induced liner fragmentation during reentry.
Fuel is sealed in containment cans to prevent contamination that could degrade semiconductor performance. Containment materials such as Mo-50%Re are chosen for low-temperature ductility. A tungsten outer coating reduces sublimation and, when roughened, can improve conversion efficiency. The containment connects to buffer layers and is lined with iridium to prevent graphite-molybdenum reactions. The outer case is aluminum alloy, with ZrO2 ceramic ball supports between the case and the containment can to reduce heat loss. Gaps are filled with layered insulation, such as 60 layers of 0.008 mm tungsten foil interspersed with ZrO2 particles. Thermophotovoltaic conversion materials are placed near the containment can to utilize up to 90% of available heat. For medical implant applications, outer casings use inert metal alloys such as platinum, tantalum, or gold and their alloys.
Applications
Aerospace
Space missions impose strict power requirements: small size, light weight, long life, and durability under harsh conditions. While solar panels are widely used for near-Earth satellites, they become impractical for deep-space missions due to weak sunlight and potential radiation damage. Radioisotope batteries meet the long-term, safe, reliable power needs of many spacecraft and have been widely adopted.
In the 20th century, many U.S. satellites, lunar modules, and deep-space probes used radioisotope power, often thermoelectric systems. For example, the 1961 Explorer 1 satellite used an isotope battery and still transmitted detectable signals in 1972. Apollo 11 carried two radioisotope units using Pu-238 fuel with 15 W thermal power; isotope batteries performed reliably in the Moon's harsh environment. Later Apollo missions used SNAP-27 RTGs with Pu-238 fuel, nominal output 63.5 W and device mass about 31 kg; actual lifetimes greatly exceeded the design life. The Cassini spacecraft used Pu-238O2 ceramic fuel pellets providing approximately 750 W at launch and about 628 W after 11 years of mission operations.
Marine, Aviation, and Navigation
Beacons and navigation buoys in remote, deep-sea, fast-current, or hazardous locations require long-lived power sources. Solar, fuel, and chemical batteries are often inadequate. Radioisotope batteries can provide decades of power without battery replacement or frequent maintenance. Militarily, they have been used to power underwater listening devices for anti-submarine surveillance, operating unattended for decades. Subsea instruments such as sonar, scientific sensors, wellhead actuators, and repeater stations can use isotope batteries that withstand depths of 5–6 km while remaining safe and cost-effective.
Remote ground installations—automatic weather stations, navigation beacons on snow-covered mountains, isolated islands, deserts, polar regions—benefit from radioisotope batteries that eliminate difficult battery replacement and maintenance.
Medical Devices
Small, lightweight, long-lived radioisotope batteries have been used in cardiac pacemakers. Thousands of patients worldwide received pacemakers powered by isotope batteries, avoiding repeated replacement surgeries. Pacemaker power units can be extremely compact and light; for example, a power source using 150 mg of Pu-238 can operate an implanted pacemaker for over 10 years. A chemical battery with equivalent lifetime would weigh on the order of an adult human. Radioisotope batteries spared patients the risk and trauma of repeated thoracic surgeries for battery replacement.
Radioisotope pacemakers were first used in the early 1970s in France and the United States. French devices used bismuth telluride semiconductor batteries with Pu-238 alloy fuel, output around 700 μW, compact dimensions, and mass about 40 g. U.S. pacemakers used metallic thermocouple batteries with metallic Pu-238 fuel, output about 230 μW. Radioisotope batteries can also power artificial hearts, artificial lungs and kidneys, cardiac assist devices, neurostimulators, blood pressure regulators, and sphincter stimulators.
Microelectromechanical Systems (MEMS)
MEMS is a rapidly developing field. Devices from automotive airbag sensors to environmental microactuators and drug-delivery systems require compact power sources. Conventional options such as fuel cells, chemical batteries, and mineral fuels are constrained by volume. Early research at Cornell University and the University of Wisconsin–Madison explored small Ni-63 sources coupled to a PN junction. Beta particles ionize the diode material, generating electron-hole pairs and producing current. Improved designs based on this principle have been developed to power small mechanical systems.
Consumer Electronics
Micro radioisotope batteries, potentially coin-cell sized, could provide extended standby times for mobile phones and other handheld electronics. Some designs considered uranium-235 as a primary component for miniature devices, aiming to provide more than one year of standby power on first use, potentially reducing the need for chargers. Microelectromechanical radioisotope batteries could also generate 10–100 V for compact RF filters or microfilters that require relatively high DC voltages.
Although technical, cost, and safety challenges remain, if these are resolved, micro radioisotope batteries could be integrated into various portable devices.
Outlook
The future of micro radioisotope batteries depends on safety, energy conversion efficiency, and cost. Minimizing the quantity of radioactive material to keep emissions low enough to be fully contained by simple packaging is key to safety. Improving conversion efficiency to increase available energy is also critical.
Another major challenge is reducing isotope cost and easing integration with electronic devices. For example, laboratory systems using 1 mCi of Ni-63 can cost about $25, which is prohibitive for mass-market electronics. Tritium may be a lower-cost alternative, produced as a byproduct in some reactors; the required amount for a micro battery could cost only a few cents. Once these hurdles are overcome, micro radioisotope batteries could find rapid adoption in handheld devices and as auxiliary power sources for conventional batteries.