Goldmann applanation tonometry
Goldmann applanation tonometry is the reference standard for measuring intraocular pressure (IOP). It is based on the Imbert–Fick law, which states that for an ideal, dry, thin-walled sphere, the internal pressure (P) equals the force (F) required to flatten its surface divided by the flattened area (A), i.e., P = F/A. Goldmann applanation tonometry measures the force required to flatten a corneal area with a 3.06 mm diameter and is relatively unaffected by ocular rigidity.
Effect of corneal factors on Goldmann readings
Corneal properties can affect Goldmann readings in several ways. Goldmann tonometers are calibrated assuming a central corneal thickness (CCT) of about 500 μm, although later studies indicate that readings most closely reflect true anterior chamber pressure when CCT is around 520 μm. Thicker corneas tend to produce artificially high IOP readings, while thinner corneas tend to produce artificially low readings. Corneal edema can produce falsely low IOP readings because IOP measurements are also affected by corneal hysteresis or relative corneal stiffness. Goldmann, Perkins, pneumotonometry, noncontact tonometry, and Tono-Pen devices are all influenced by CCT, and there is no universally accepted correction factor for CCT with applanation tonometry.
Rebound tonometer operating principle
The rebound tonometer uses a magnetized steel probe with a plastic-coated shaft that is propelled toward the cornea by a pulsed electromagnetic coil. When the probe strikes the cornea it rebounds back into the instrument, producing a voltage change. A coil inside the tonometer detects the voltage change; the generated voltage is proportional to the probe's rebound velocity. Higher IOP produces a shorter contact time and a faster rebound velocity. The probe motion induces magnetic field changes that are interpreted as IOP on the device display. Rebound tonometers generally do not require regular calibration. Probes are single-use, 24 mm long, weighing about 11 mg; the probe tip diameter is 1 mm and is rounded to minimize corneal injury risk.
Rebound tonometer readings are frequently higher than Goldmann values, particularly at IOPs above 22 mm Hg. On average, rebound measurements are 2.0–3.1 mm Hg higher than Goldmann applanation measurements (range: 5 to 12 mm Hg), and this difference becomes more pronounced with increased CCT. In approximately 75% of comparisons, rebound tonometer values exceed Goldmann values.
Instrument components
A rebound tonometer consists primarily of the probe, a track containing a coaxial coil, and a signal acquisition unit.
Measurement sequence
- When loaded, the probe is first magnetized by an internal permanent magnet.
- When the coaxial coil is energized, it generates a magnetic field that propels the probe outward.
- After the probe contacts the cornea its forward velocity decreases, producing a negative acceleration.
- The magnitude of that acceleration is measured by the signal acquisition device.
- The measured acceleration is correlated with IOP and converted to an IOP value using a regression equation.
Importance of probe alignment
For the signal receiver to correctly detect the acceleration generated by corneal rebound, the probe must be aligned perpendicular to the corneal apex. This alignment ensures the corneal reaction force produces acceleration directed toward the receiver. Misalignment from the corneal apex can cause substantial error. If the probe does not strike the corneal vertex, the curvature causes an off-axis, tangential rebound that produces torque, so the probe does not rebound directly backward, producing a larger measurement error.
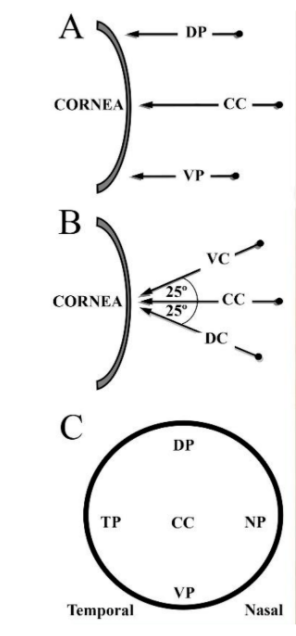
Note: the following material is from a veterinary publication illustrating probe positioning on a canine cornea.
Corneal positions and definitions
Illustrations show probe positioning on the canine corneal surface:
- A: Peripheral positions in vertical view, showing displacement from central cornea (CC) to dorsal peripheral (DP) and ventral peripheral (VP).
- B: Angular positions in vertical view, showing ventro-central (VC) and dorso-central (DC).
- C: Frontal view of peripheral positions, showing displacement from central cornea (CC) to dorsal (DP), ventral (VP), nasal (NP), and temporal (TP).
Study: positional variability of rebound tonometer on canine cornea
Previous studies have shown that rebound tonometer readings obtained at different corneal positions lack agreement with readings obtained at the central corneal apex. This study assessed the variability of rebound tonometer IOP readings taken on peripheral and angled positions of the canine cornea.
Forty-six client-owned dogs presenting for ophthalmic examination at the Queen's Veterinary School Hospital, University of Cambridge, were included. IOP was measured at different positions and angles on the cornea:
- Perpendicular to the corneal center (central cornea, CC);
- At the corneal center with the tonometer at four different angles;
- At four peripheral corneal points.
All values were compared to the recommended CC position values.
Results
IOP values at seven noncentral positions were significantly underestimated. Median and interquartile ranges varied from 12.1 ± 4 mm Hg (nasal peripheral) to 15 ± 5 mm Hg (central lateral angle), with differences from the CC value ranging from 0 to 2.9 mm Hg. The dorsal central cornea did not differ significantly from CC (p = 0.09). Peripheral positions produced lower median readings than angled central positions. These results indicate that tilting the tonometer or measuring peripheral regions can cause small but statistically significant underestimation of IOP.
These findings suggest the need for a reliable positioning system to ensure consistent probe placement.
Probe-to-cornea distance study in dogs and rats
A separate study examined the effect of probe-to-cornea distance on IOP measurements in dogs and rats. Subjects included 24 conscious dogs and 15 anesthetized Wistar rats. Methods: three interchangeable 3D-printed polylactic acid (PLA) spacer rings replaced the original tonometer spacer to provide probe-to-cornea distances of 4, 6, and 8 mm. IOP readings were obtained sequentially at probe distances of 4, 6, and 8 mm by a single observer. Dogs were lightly restrained; rats were anesthetized with isoflurane.
Results and conclusion
IOP values at probe-to-cornea distances of 4, 6, and 8 mm differed significantly for both dogs and rats (P < 0.01). There was a small positive correlation between IOP (mm Hg) and probe-to-cornea distance (mm): dogs r = 0.39, rats r = 0.51. Mean IOPs for dogs were 16.2 ± 3.0 mm Hg at 4 mm, 17.6 ± 3.4 mm Hg at 6 mm, and 19.8 ± 3.8 mm Hg at 8 mm. For rats, mean IOPs were 8.2 ± 1.5 mm Hg at 4 mm, 9.4 ± 1.8 mm Hg at 6 mm, and 10.5 ± 1.5 mm Hg at 8 mm. Conclusion: probe-to-cornea distance significantly affects IOP readings, even within the manufacturer-recommended 4–8 mm range.