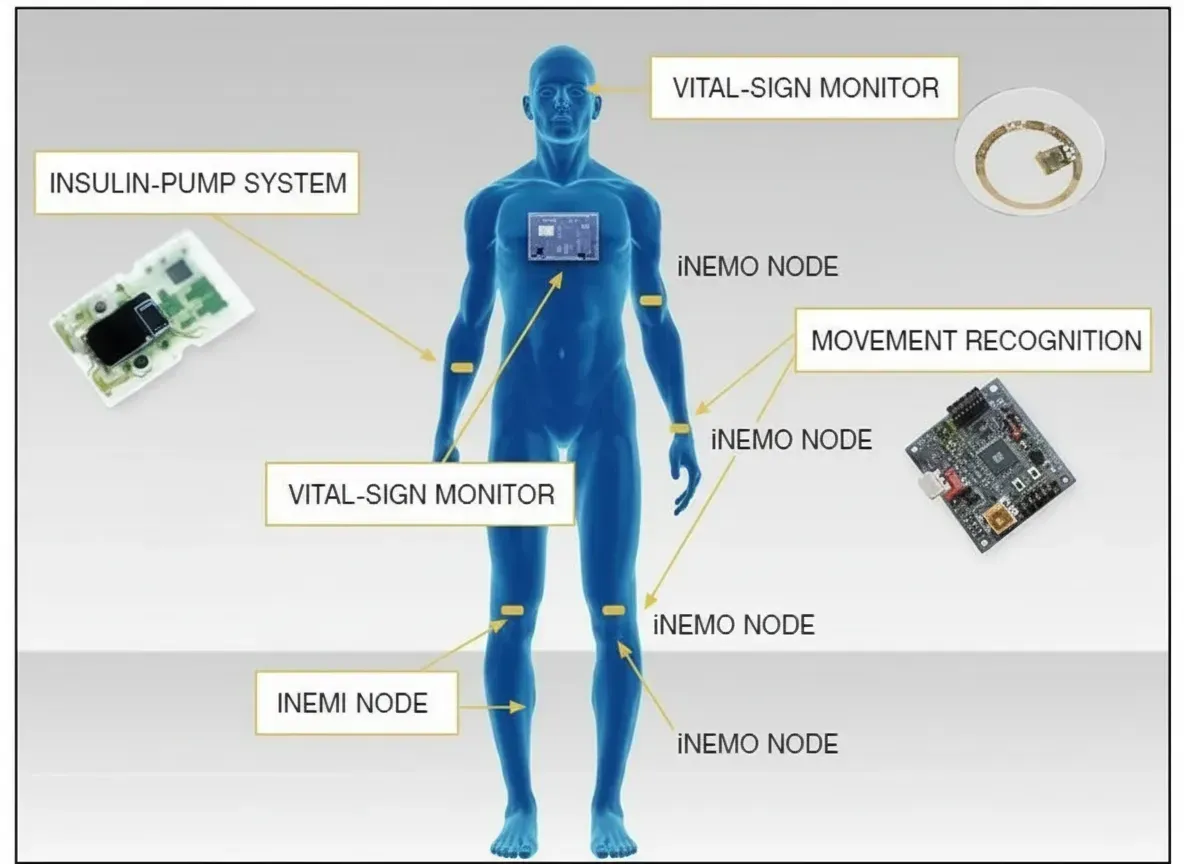
Overview
Metal oxysulfides are an emerging class of sensing materials for high-performance nitrogen dioxide (NO2) detection because they can operate at room temperature, show strong selectivity to NO2, achieve detection limits at the parts-per-billion level, and exhibit high environmental stability.
According to MIMS Consulting, researchers at RMIT University investigated the room-temperature NO2 sensing performance of three-dimensional self-assembled zinc oxysulfide made from ultrathin nanosheets. Under 460 nm excitation, the sensor exhibited a fully reversible and repeatable response to 1.26 ppm NO2 at room temperature, with a response magnitude of about 2.27 and a limit of detection (LOD) of 294.8 ppt. The study was published as "A Room Temperature High-Performance Visible-Light-Assisted NO2 Gas Sensor Based on Ultrathin Zinc Oxysulfide" in the journal Advanced Sensor Research.
Synthesis and Structure
The material was prepared by a combined hydrothermal and annealing route. Zinc sulfide microself-assemblies were synthesized first and then converted to zinc oxysulfide under a controlled environment. The resulting zinc oxysulfide formed mixed spherical 3D structures with sizes in the 5–50 μm range. Most sulfur atoms in the original zinc sulfide were replaced by oxygen, causing the crystal structure to change from cubic/hexagonal to an orthorhombic configuration. The optical bandgap decreased from about 3.6–3.8 eV to approximately 1.92 eV, enabling visible-light absorption.
Material Characterization
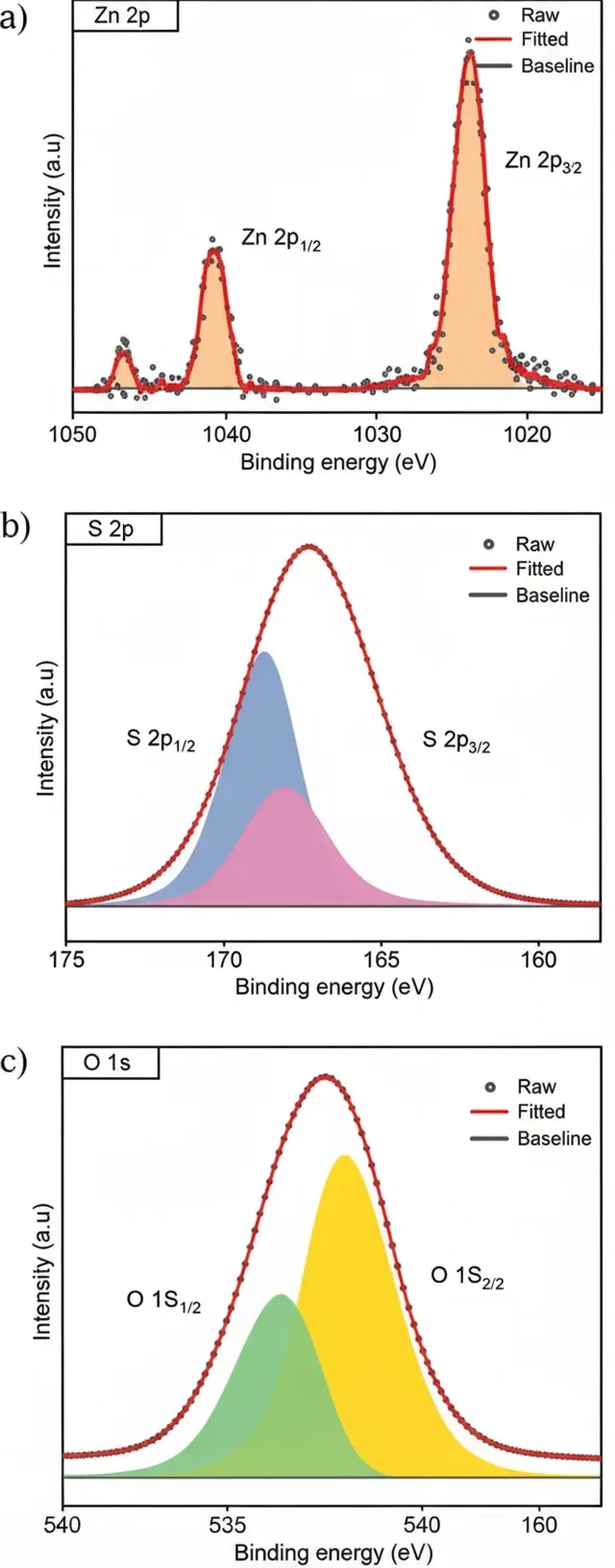
The zinc oxysulfide was characterized by X-ray photoelectron spectroscopy (XPS) to analyze its chemical composition and bonding states.
Sensor Fabrication and Performance
Sensors were fabricated by depositing approximately 40 μL of the material solution onto silicon interdigitated transducer (IDT) substrates. NO2 was selected as the target gas due to its paramagnetic character. Under blue-light illumination (λ = 460 nm), the zinc oxysulfide sensor produced a fully reversible response to 1.26 ppm NO2, with a response factor near 2.27. The response is mainly attributed to physical adsorption between gas molecules and the sensing material. The calculated LOD is 294.8 ppt, placing this device among the top-performing semiconductor-based chemiresistive sensors. The sensor also displayed high selectivity for NO2 compared with other analytes and showed only minor performance degradation when exposed to varying humidity.
Summary
The researchers synthesized ultrathin zinc oxysulfide nanosheets via a two-step chemical route. Due to self-assembly and aggregation, the material forms 3D spherical structures. Chemical and crystallographic analysis indicates a ZnSO4-type lattice containing Zn, S, and O bonds. Strong optical absorption and significantly extended exciton radiative lifetimes enabled room-temperature, visible-light-assisted gas sensing on a light-gas-material interaction platform. Under blue-light excitation (λ = 460 nm), the sensor showed a fully reversible and repeatable response to 1.26 ppm NO2 at room temperature, with a response magnitude of about 2.27. Dynamic tests demonstrated linear sensor behavior from low to high NO2 concentrations, with an LOD of 294.8 ppt. The device also responded relatively quickly to low-concentration gas, requiring roughly 8 minutes to fully respond to 315 ppb NO2, and exhibited greater selectivity to NO2 than to gases such as H2, H2S, CO2, CH4, and NH3. The authors conclude the work highlights the potential of ultrathin metal oxysulfides for next-generation, room-temperature, visible-light-assisted gas sensors.