Research background
Lithium metal offers extremely high specific capacity (3860 mAh g^-1) and the lowest redox potential (?3.04 V versus SHE), making it an attractive anode material for next-generation rechargeable batteries. However, challenges including low Coulombic efficiency (CE), short cycle life, and uncontrolled lithium dendrite growth limit the commercialization of lithium metal batteries (LMBs). Concentration gradients near the lithium surface are a primary driver of dendrite formation; addressing this issue is critical for realizing high-performance lithium metal cells.
Summary of results
Researchers led by Wei-Hong Lai at the University of Wollongong developed a multilayer crystalline coating of electroactive β-phase PVDF-HFP implanted with lithium nitrate (LiNO3), referred to as PHL. Negatively charged polymer chains on the surface adsorb Li+, forming charged lithium-ion channels. These channels act as local reservoirs that continuously release lithium ions to compensate electrolyte ion flux, thereby reducing dendrite growth.
The approach achieved a Li||Cu cell Coulombic efficiency of up to 97.0% over 250 cycles, and stable symmetric plating/stripping behavior for 2000 hours at 3 mA cm^-2 with lithium utilization up to 50%. Full cells using PHL-Cu@Li anodes paired with LiFePO4 cathodes showed long-term cycling stability, retaining 95.9% capacity after 900 cycles. Notably, full cells paired with LiNi0.87Co0.1Mn0.03O2 retained 170.0 mAh g^-1 discharge capacity and 84.3% capacity retention after 100 cycles under an aggressive N/P ratio of 0.83. The work was published as "Lithium Ion Charged Polymer Channels Flattening Lithium Metal Anode" in Nano-Micro Letters.
Highlights
- LiNO3-implanted electroactive β-phase PVDF-HFP was constructed as an artificial solid electrolyte interphase (SEI) layer to suppress dendrite growth.
- Negatively charged polymer layers capture Li+ on their surfaces, forming charged lithium-ion channels that serve as lithium reservoirs and compensate electrolyte ion supply, providing a continuous source of Li+.
- The modified lithium anode achieved over 2000 hours of long cycling life in symmetric cells at an ultrahigh lithium utilization (50%), and full cells operated for 100 cycles under demanding conditions, including an N/P ratio of 0.83.
Figure guide
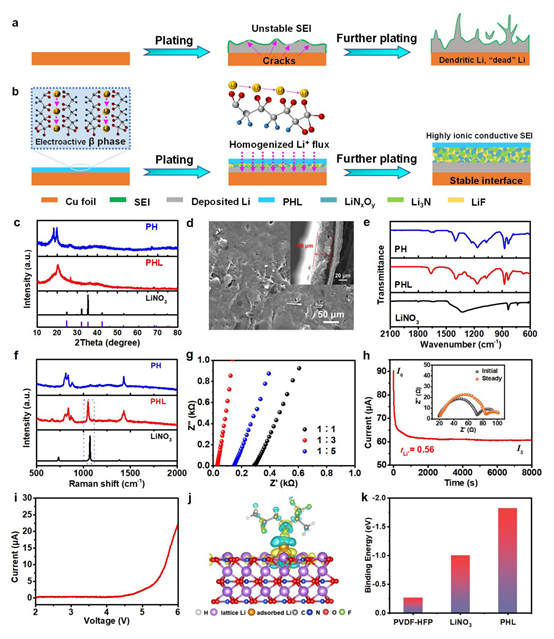
Figure 1: a Schematic of lithium plating/stripping on bare Cu and b PHL-Cu electrodes; c XRD patterns of LiNO3, PH and PHL; d SEM image of PHL-Cu electrode; e FTIR and f Raman spectra of PH, PHL and LiNO3; g chronoamperometry and corresponding EIS of PHL-Cu@Li before and after polarization; h LSV of a PHL film (mass ratio 3:1) in Li||SS cell at a scan rate of 0.2 mV s^-1; i EIS of SS|PHL|SS cells with different mass ratios at room temperature; j charge density distribution of PHL at different charge states; k binding energies of Li with PVDF-HFP, LiNO3 and PHL.
Materials and structural characterization
PHL was prepared by loading PVDF-HFP (PH) with different mass fractions of LiNO3. XRD comparison of PH, PHL and LiNO3 shows a reduced intensity of the nonpolar α phase in PHL, indicating that the introduced LiNO3 promotes formation of the electroactive β phase, likely due to ion–dipole interactions between ions and the polymer matrix. SEM images show that the PHL composite film has a relatively smooth surface with a thickness of about 36 μm, unlike the porous PH structure, confirming full integration of LiNO3 with the polymer matrix. FTIR and Raman spectra further characterize structural features: FTIR peaks at 837, 874 and 1275 cm^-1 correspond to the polar β phase; new peaks at 675 and 1664 cm^-1 in PHL likely arise from crosslinking caused by interaction between Li+ and polar functional groups in PH. Raman peaks at 800 and 837 cm^-1 correspond to α and β phase modes; the α-phase intensity decreases after LiNO3 introduction. A broader peak at 1435 cm^-1 indicates β-phase activity and reduced crystallinity. The LiNO3-related peak at 1070 cm^-1 slightly shifts to 1043 cm^-1 after integration with the polymer, suggesting interaction between -CF- groups and Li+.
Ion conductivity and electrochemical properties
Ionic conductivity is a key metric for artificial SEI layers, affecting Li+ transport and uniform deposition. EIS tests on assembled SS|PHL|SS cells at room temperature indicate an ionic conductivity of 3.39e-4 S cm^-1 for the PHL composite layer at a mass ratio of 1:3, a high value that favors uniform lithium deposition at high current densities. Chronoamperometry and EIS before and after polarization show that PHL-Cu@Li has a Li+ transference number (tLi+) of 0.56, significantly higher than bare lithium (0.27). The high tLi+ results from crosslinking between Li+ and -CF- functional groups, which prevents strong electric fields caused by anion depletion and thereby suppresses dendrite growth. LSV demonstrates electrochemical stability: the PHL film (mass ratio 3:1) shows a high anodic stability (>5.25 V), indicating reliability under high-voltage operation. DFT calculations of Li adsorption on PHL components reveal pronounced charge transfer consistent with strong Li–PHL interactions. Calculated binding energies increase in the order: PVDF-HFP (?0.26 eV) < LiNO3 (?1.00 eV) < PHL (?1.82 eV), indicating higher Li affinity and more Li diffusion channels in PHL compared with PH or LiNO3 alone, which facilitates rapid Li+ transport and uniform deposition.
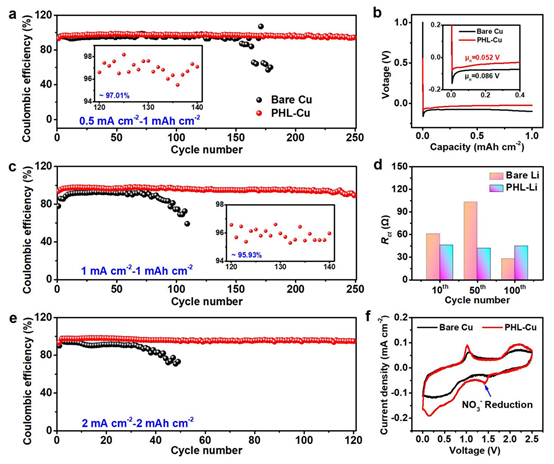
Figure 2: a CE and b initial plating voltage curves of Cu or PHL-Cu electrodes in Li||Cu cells at 0.5 mA cm^-2 and 1 mAh cm^-2; c CE at 1 mA cm^-2 and 1 mAh cm^-2; d Rct evolution with cycle number; e CE of PHL-Cu electrode at 2 mA cm^-2 and 2 mAh cm^-2; f CV curves of Li||Cu cells with Cu or PHL-Cu electrodes from 0 to 2.5 V at 5 mV s^-1.
Electrochemical cycling on Cu and PHL-Cu current collectors
Li||Cu cells with Cu and PHL-Cu electrodes were assembled. At 0.5 mA cm^-2 and 1 mAh cm^-2, the PHL-Cu electrode maintained stable cycling for 250 cycles with CE around 97.0%, whereas bare Cu showed lower CE and shorter life. Initial plating curves show a lower nucleation overpotential for PHL-Cu (52 mV) compared with Cu (86 mV), indicating lower nucleation barrier and faster kinetics on PHL-Cu. At 1 mA cm^-2, cells with PHL-Cu remain stable beyond 250 cycles with an average CE of 95.9%, outperforming bare Cu (which decayed to 80% within 100 cycles).
To show the advantage of the PHL layer, 0.257 M LiNO3 (matching the PHL layer) was added to the ester electrolyte as an additive. Compared with bare Cu, PHL-Cu exhibits lower interfacial impedance after cycling, indicating more favorable plating/stripping kinetics. Even at 2 mA cm^-2 under equal capacity conditions, the PHL-Cu cell maintained stable cycling for 120 cycles with CE up to 95.4%, while the bare Cu cell decayed after ~30 cycles. CV studies indicate that PHL-Cu@Li cells show a broad peak at 1.42 V attributed to LiNO3 reduction; the decomposition products are nitrogen-containing species that promote fast Li+ transport and uniform distribution.
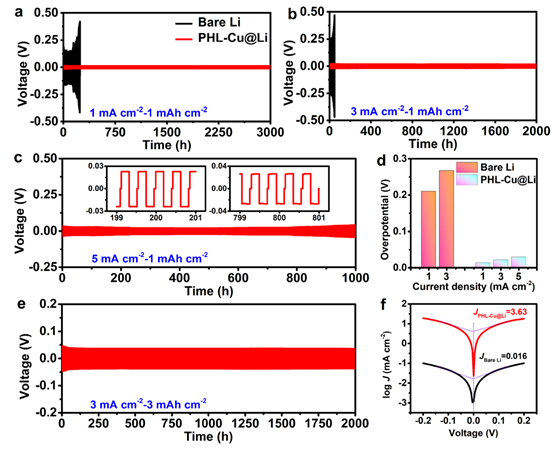
Figure 3: a–c Voltage profiles of symmetric Li||Li cells using bare Li or PHL-Cu@Li at 1, 3 and 5 mA cm^-2 under fixed capacity of 1 mAh cm^-2; d overpotentials of bare Li and PHL-Cu@Li symmetric cells; e PHL-Cu@Li symmetric cell voltage at 3 mA cm^-2 and 3 mAh cm^-2; f Tafel curves for Li||Li symmetric cells within ?0.2 to 0.2 V.
Symmetric cell performance and lithium utilization
Symmetric cell constant-current tests under different current density and capacity conditions were performed. A PHL-Cu@Li electrode was prepared by plating 6 mAh cm^-2 Li onto PHL-Cu at 0.5 mA cm^-2. In ester electrolyte, Li||Li symmetric cells with bare Li exhibited increasing overpotentials and failure at 255 hours (1 mA cm^-2) and 50 hours (3 mA cm^-2), indicating interfacial fluctuations caused by dendrites and "dead Li". In contrast, cells with PHL-Cu@Li showed long lifetimes with low overpotentials: 3000 hours at 1 mA cm^-2, 2000 hours at 3 mA cm^-2, and 1000 hours at 5 mA cm^-2.
The excellent plating/stripping stability at low overpotential is attributed to a stable electrode/electrolyte interface: LiNO3 preloaded in the polymer framework continuously decomposes into nitrogen-containing species, improving SEI stability during operation. The electroactive β phase with abundant functional groups crosslinks with Li+, further stabilizing the interface and accelerating Li+ transport. Under the protective layer, PHL-Cu@Li anodes show excellent cycling stability over 2000 hours and fast Li+ transport; at 3 mA cm^-2, lithium utilization reached 50%. At a capacity of 4 mAh cm^-2 (66.7% lithium utilization), PHL-Cu@Li still exhibited excellent stability over 2000 hours. The PHL-Cu@Li anode maintained stable plating/stripping with small hysteresis for 1000 hours. Tafel analysis indicates the exchange current density of PHL-Cu@Li remained stable over 1000 hours with low hysteresis; the exchange current density (3.63 mA cm^-2) is far higher than that of bare Li (0.016 mA cm^-2), indicating faster charge transfer consistent with time-dependent and EIS results.

Figure 4: a,d Schematics of lithium plating on Cu and PHL-Cu electrodes; b,c SEM images of Cu after 50 cycles at 1 mA cm^-2; e,f SEM images of PHL-Cu after 50 cycles at 1 mA cm^-2; g,h in situ optical microscopy of Li plating at high current density (10 mA cm^-2) on Cu and PHL-Cu.
Morphology evolution and in situ observation
Evolution of lithium deposition morphology on Cu and PHL-Cu electrodes was studied. For Cu, inherent protrusions and cracks lead to local Li+ flux concentration and uneven deposition, producing "dead Li" layers and rapid electrolyte consumption. With a PHL layer, uniform, dendrite-free lithium deposition is achieved because: (1) the polymer matrix acts as a reservoir that releases LiNO3 gradually, forming nitrogen-containing species that inhibit dendrite growth; (2) the PHL layer provides abundant polar functional groups and high ionic conductivity to homogenize Li+ flux and promote uniform plating/stripping. SEM after 50 cycles at 1 mA cm^-2 and 1 mAh cm^-2 shows the Cu surface is rough with abundant dendrites and a ~9 μm dead Li layer, while PHL-Cu remains smooth with no dendritic protrusions and uniform Li deposition within the PHL, demonstrating that the robust PHL layer homogenizes Li+ flux and accelerates interfacial ion transport to form compact, dendrite-free deposits.
In situ optical microscopy at 10 mA cm^-2 on Li||Cu cells with bare Li counter electrodes shows that bare Cu develops dendrites within 1 minute of deposition and accumulates dead Li and dendritic lithium over time due to a weak SEI. In contrast, PHL-Cu displays uniform, smooth deposition throughout the experiment, with no observable dendrites or dead Li, indicating that the PHL layer guides uniform lithium deposition and reduces parasitic interfacial reactions.
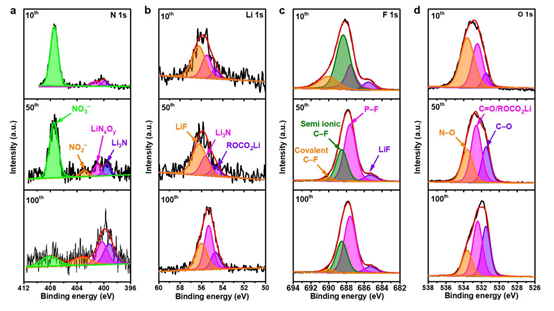
Figure 5: XPS spectra of SEI on PHL-Cu electrodes after different cycles in Li||Cu cells: a N 1s, b Li 1s, c F 1s and d O 1s.
SEI composition analysis
XPS was used to probe elemental composition on the Cu electrode. N 1s spectra show peaks at 399.6, 400.8, 403.2 and 407.5 eV assigned to Li3N, LiNxOy, NO2^- and NO3^-, originating from LiNO3 decomposition. With continued cycling, the contents of Li3N, LiNxOy and NO2^- increase while NO3^- decreases, indicating the polymer framework acts as a reservoir that gradually releases LiNO3 into the electrolyte. Li 1s spectra show distinct peaks at 54.9, 55.5 and 56.0 eV attributed to ROCO2Li, Li–N and LiF; the Li–N signal increases with cycling, further indicating continuous formation of Li3N and LiNxOy. F 1s spectra show peaks at 685.3, 687.5, 688.3 and 690.2 eV associated with LiF, P–F, semi-ionic C–F and covalent C–F bonds. In O 1s spectra, PHL-Cu shows an additional N–O peak at 533.6 eV compared with Cu, further confirming generation of nitrogen-containing species. Nitrogen-containing SEI species formed from LiNO3 reduction improve SEI stability and ionic conductivity, suppress dendrites and accelerate Li+ transport to enable uniform lithium deposition.
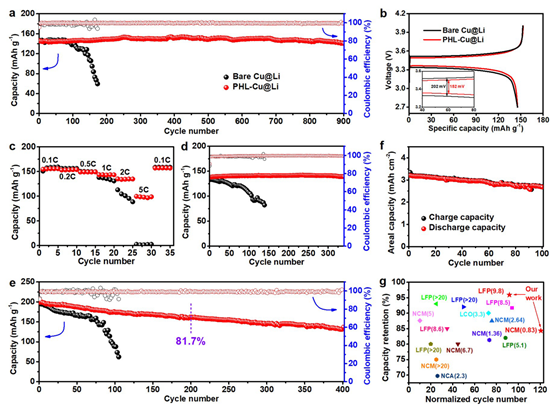
Figure 6: a Cycling stability of Cu@Li||LFP and PHL-Cu@Li||LFP full cells at 0.5C; b initial charge/discharge curves; c rate performance; d cycling stability at 1C; e cycling stability of Cu@Li||NCM and PHL-Cu@Li||NCM at 0.5C; f PHL-Cu@Li||NCM full cell cycling at ultra-low N/P ratio of 0.83; g comparison of capacity retention and cycle life of PHL-Cu@Li against practical LMB benchmarks.
Full-cell performance with commercial cathodes
To evaluate practical value, full cells were assembled with Li anodes and commercial cathodes (LFP or NCM). Cu@Li and PHL-Cu@Li anodes were prepared by depositing defined amounts of Li onto Cu and PHL-Cu at 0.5 mA cm^-2. At 0.5C (1C = 170 mAh g^-1, N/P = 9.8), PHL-Cu@Li||LFP cells cycled for 900 cycles with 95.9% capacity retention and an average CE of 99.7%. In contrast, Cu@Li||LFP cells showed poor performance within 200 cycles, retaining only 40.1% capacity with lower CE. Initial charge/discharge curves in the 2.7–4.0 V window show similar discharge capacities for PHL-Cu@Li and Cu@Li (145.7 vs. 145.6 mAh g^-1) but lower polarization for the PHL-modified cell, indicating faster Li+ transport. In rate tests, PHL-Cu@Li delivers higher discharge capacity at high rates; at 1C and 2C, it maintained excellent cycling for 340 and 150 cycles, respectively.
PHL-Cu@Li||NCM full cells had an initial capacity of 198.9 mAh g^-1 at 1C (1C = 200 mAh g^-1) and retained 68.5% capacity after 400 cycles (81.7% after 200 cycles) with an average CE of 99.1%. Cu@Li||NCM cells showed much lower capacity and CE after 110 cycles. Impressively, a limited PHL-Cu@Li anode (3 mAh cm^-2) paired with a high-loading NCM cathode (18 mg cm^-2) at N/P = 0.83 maintained 100 cycles with 84.3% capacity retention at 0.5C. The cycling stability and capacity retention of PHL-Cu@Li full cells compare favorably with many reported results. The extended cycle life of Li||LFP and Li||NCM full cells is attributed to the PHL layer acting as a LiNO3 reservoir that forms a stable, nitrogen-containing SEI that stabilizes the electrode/electrolyte interface and accelerates Li+ transport, resulting in uniform lithium deposition and dendrite suppression.
Conclusions and outlook
LiNO3-implanted electroactive β-phase PVDF-HFP multilayer coatings form charged lithium-ion channels on the lithium surface. Electroactive polymer chains capture Li+ and create charged channels that act as local Li reservoirs. Nitrogen-containing ionic conductive species produced by LiNO3 decomposition, together with extended molecular channels, homogenize Li+ flux and facilitate ion transport, substantially suppressing dendrite growth and enabling uniform lithium deposition. In ester electrolytes, Li||Cu cells achieved 97.0% CE over 250 cycles, and symmetric cells showed stable performance at high current density and high lithium utilization. Full cells with PHL-Cu@Li anodes paired with LFP or NCM cathodes demonstrated significantly extended cycle life and improved capacity retention compared with bare Li anodes, including strong performance under an aggressive N/P ratio of 0.83.