Overview
Real-time detection of biomolecules is important for in vitro diagnostics, early disease detection, and routine health monitoring. Steroid hormones such as progesterone (P4), estradiol (E2), and testosterone exist at very low concentrations in the body (< nM) but play critical roles in growth, development, and reproduction. Clinical hormone assessment typically relies on invasive blood sampling and complex laboratory equipment operated by trained personnel. Noninvasive saliva testing is an attractive alternative, but free hormones in saliva represent only about 1% to 2% of total blood hormone levels, which limits practical clinical use. There is therefore a large need for hormone assays with ultra-low detection limits, wide dynamic range, and simple operation for dynamic monitoring of sex hormones.
Sensor concept
Researchers from the School of Materials Science and Engineering, Sun Yat-sen University, developed a three-dimensional microstructured flexible conducting polymer electrode to achieve low-cost, sub-femtomolar electrochemical immunosensing of sex steroid hormones. The sensor can directly detect highly diluted saliva samples without centrifugation, enabling at-home saliva testing and monitoring of salivary P4 and E2 across the menstrual cycle.
Electrode design
The sensor core is a conductive polymer electrode based on micro-pyramidal arrays coated with PEDOT:PSS doped with lithium bis(trifluoromethanesulfonyl)imide (LiTFSI), combined with gold nanoparticles (AuNPs) to facilitate immobilization of hormone antibodies. The simple immunosensing working electrode uses microcone tips to promote molecular diffusion and enrichment. Using differential pulse voltammetry (DPV), the sensor detects hormones by the decrease in peak current, achieving sensitive electrochemical detection of P4 and E2 from sub-femtomolar to micromolar concentrations within minutes.
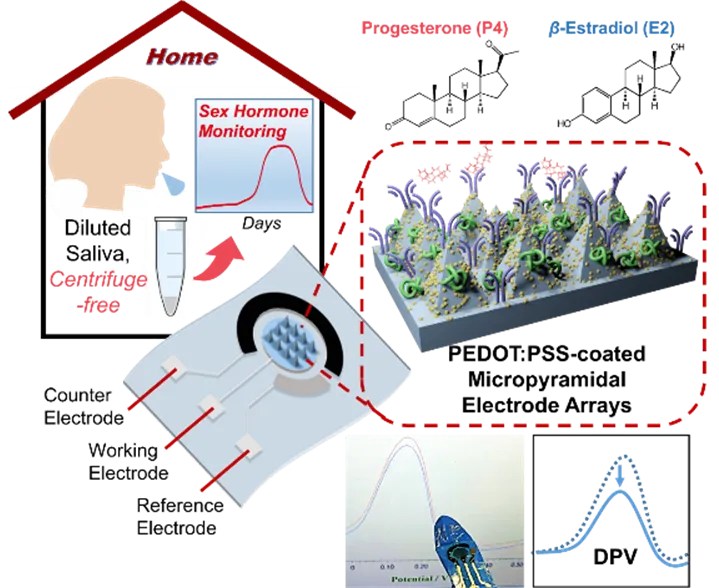
Figure 1. Schematic of a PEDOT:PSS-coated flexible micro-pyramid array used as the working electrode for electrochemical immunosensing of salivary progesterone (P4) and beta-estradiol (E2). The flexible wearable device uses differential pulse voltammetry (DPV) for sensitive detection of steroid hormones.
Microstructure and electrochemical simulation
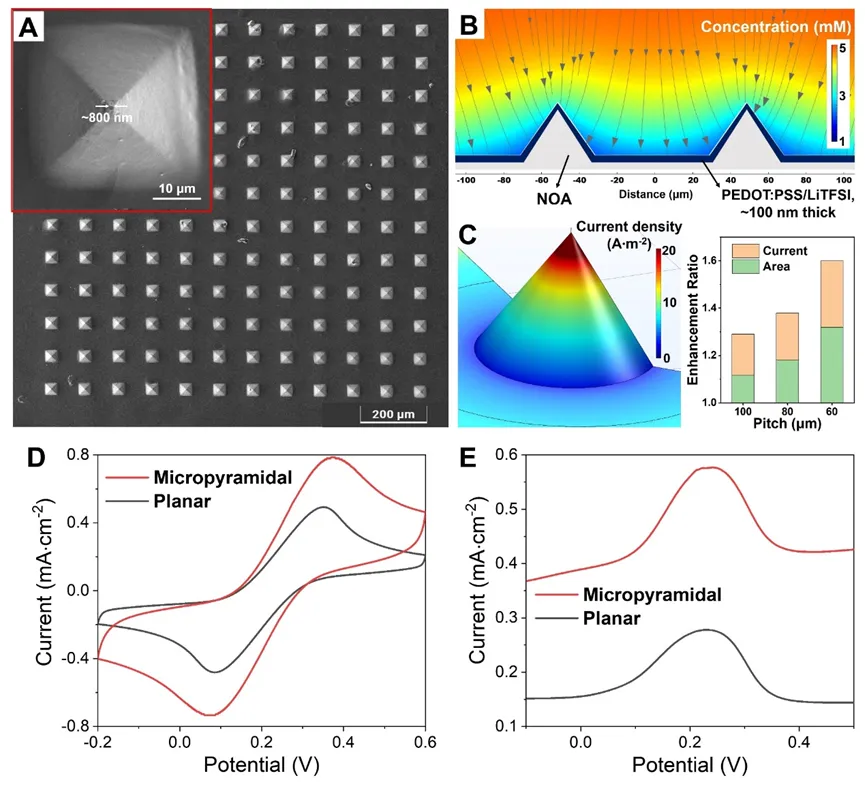
Figure 2. (A) SEM image of micro-pyramid arrays coated with PEDOT:PSS/LiTFSI. (B) Finite element simulation of diffusion profiles of Fe(CN)6 4- in the electrolyte around the micro-pyramid electrode array under an overpotential. (C) Three-dimensional local current density distribution for a single micro-pyramid electrode and the enhancement of current and area with size variation. (D–E) Comparison of cyclic voltammetry (CV) and DPV current responses between micro-pyramid arrays and planar PEDOT:PSS/LiTFSI electrodes.
Immunosensor fabrication and analytical performance
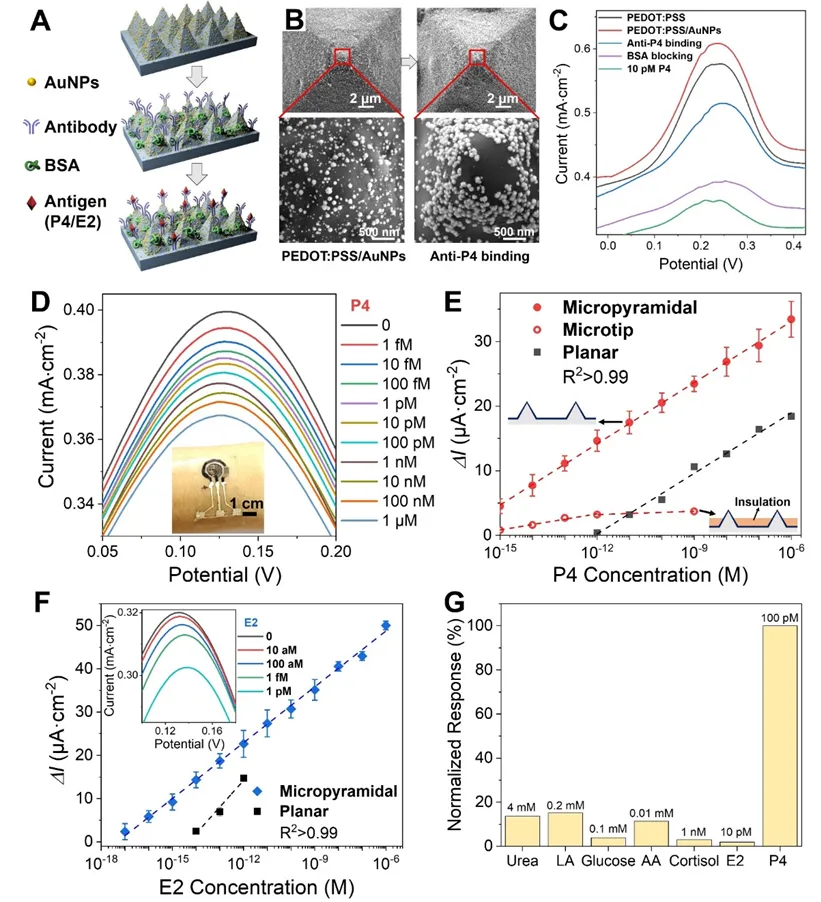
Figure 3. (A) Schematic of immunosensor fabrication. (B) SEM images after electrodeposition of AuNPs (left) and after incubation with P4 antibody (right) on the micro-pyramid PEDOT:PSS electrode. (C) DPV changes corresponding to electrode modification steps. (D) Typical DPV results for P4 over a detection range from 1 fM to 1 μM. (E) Calibration curves showing DPV peak current change (ΔI) versus P4 concentration for micro-pyramid, microneedle-tip, and planar electrodes. (F) DPV curves and calibration for an E2 antibody-modified immunosensor. (G) Normalized DPV responses on the immunosensor for common interferents at physiological saliva concentrations, demonstrating good selectivity.
Mechanical stability and wearable testing
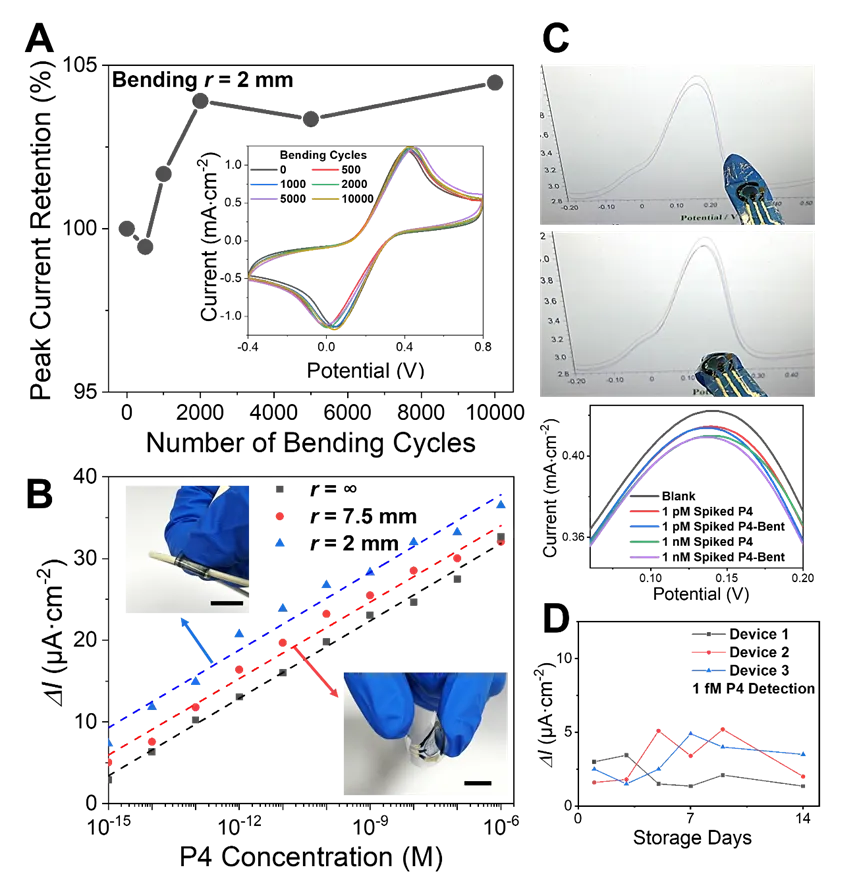
Figure 4. (A) The CV peak current of the micro-pyramid electrode remained essentially unchanged after 10,000 repeated bending cycles at a bending radius of 2 mm; inset shows representative CV curves during bending cycles. (B) Standard calibration curves for P4 detection under different bending conditions. All scale bars are 1 cm. (C) The immunosensor mounted on a glove for P4 testing showed negligible interference from finger bending; corresponding DPV peak current changes were negligible. (D) After storage at 4°C for 14 days, the device still detected 1 fM P4.
At-home, centrifuge-free saliva testing
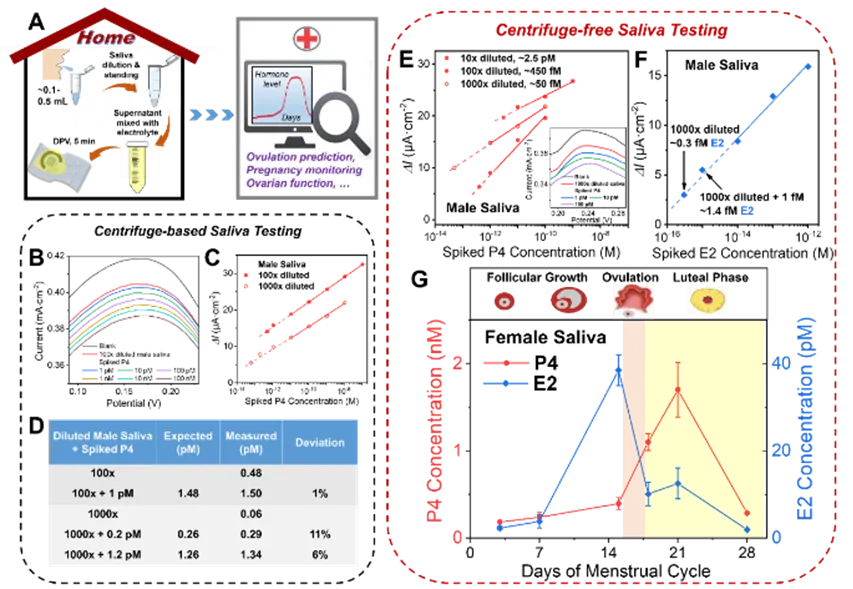
Figure 5. (A) Schematic of a centrifuge-free at-home saliva hormone testing workflow for female health monitoring. (B–D) Results from male saliva samples after centrifugation, including DPV curves for P4 spiked into 100-fold diluted saliva and calibration curves and accuracy for 100-fold and 1000-fold diluted samples. (E) Centrifuge-free testing: DPV responses for a series of P4 spikes in 10- to 1000-fold diluted male saliva, used to determine the sample background P4; inset shows the DPV for a 1000-fold diluted sample. (F) Calibration for E2 spiked into 1000-fold diluted male saliva. (G) Centrifuge-free saliva testing of a 1000-fold diluted female saliva sample monitored over 4 weeks for P4 and E2 levels. Hormone level changes corresponded to menstrual cycle phases and enabled detection of a short ovulation window.
Summary and potential applications
This work demonstrates a flexible electrochemical immunosensor using PEDOT:PSS/AuNPs-coated 3D micro-pyramid electrodes for sub-femtomolar detection of steroid hormones. The 3D microstructured electrode enhances mass transport to lower the limit of detection by roughly three orders of magnitude while maintaining a wide linear range from sub-femtomolar to micromolar concentrations. Such low detection limits are well suited to measuring ultra-low concentrations of steroid hormones in highly diluted human saliva, enabling noninvasive, centrifuge-free at-home monitoring of female hormone levels.
The proposed 3D micro-pyramidal polymer electrode concept for improving detection limits and dynamic range could be explored as a general biosensing approach. From precise clinical diagnostics and in vivo detection of bioactive species to trace monitoring of contaminants in food and the environment, this ultrasensitive and mechanically compliant 3D microstructured electrode platform could support a range of applications.
Publication and funding
The work was published as "Tip-Enhanced Sub-Femtomolar Steroid Immunosensing via Micropyramidal Flexible Conducting Polymer Electrodes for At-Home Monitoring of Salivary Sex Hormones" in ACS Nano. Zhaoxian Li, a master’s student at the School of Materials Science and Engineering, Sun Yat-sen University, is the first author. Associate Professor Zhuang Xie (School of Materials Science and Engineering) and Dr. Limei Zhang (Reproductive Medicine Center, The First Affiliated Hospital of Sun Yat-sen University) are corresponding authors.
The research was supported by the National Natural Science Foundation of China (22075325), Guangzhou Science and Technology Bureau (202002030434, 2023A04J2177), and Sun Yat-sen University Key Cultivation Project (23ptpy51).