Introduction
When a mobile phone enters an elevator, the signal often drops sharply because the metal elevator acts as a Faraday cage. Could aluminum foil produce a similar signal shielding effect?
Q1: Why does high-voltage transmission reduce power loss?
Electric power loss in transmission mainly comes from Joule heating, which is caused by the resistance of the conductors. Joule heating power is given by P = I2R, so to reduce losses you can either lower the conductor resistance or reduce the current. For a given transmitted power, raising the transmission voltage reduces current (since P = VI), hence high-voltage transmission reduces I and thus reduces resistive losses.
Raising system voltage introduces other issues such as insulation, radiation, and safety concerns. For that reason, high-voltage lines are often routed through sparsely populated areas and are carried on towers to ensure safety.
Q2: Why are solid iron blocks silver-white while iron powder is black?
The high reflectivity of an iron block comes from its good electrical conductivity. Electromagnetic waves penetrate only a very small distance into a conductor (the skin depth), so visible light is mostly reflected from a smooth metal surface, producing a silver-white metallic luster.
Iron powder, with particle sizes on the order of 1–100 μm, appears dark or black for three main reasons. First, powder lacks a smooth surface, so visible light undergoes diffuse reflection rather than specular reflection. Second, powder particles have higher chemical reactivity and typically carry complex oxide layers on their surfaces; these dark oxide layers absorb visible light more strongly. Third, the large surface area and porous packing of the powder cause multiple internal reflections, further reducing the net reflectance. The combined effect makes iron powder appear dark.
Q3: If a phone is completely wrapped in aluminum foil, will it be isolated from signals?
This is an electrodynamics question. When electromagnetic waves strike a metal surface, some energy is reflected and some penetrates into the metal and is absorbed.
Most metals absorb electromagnetic waves well once the waves penetrate the surface. For time-varying fields (electromagnetic waves), the field amplitude decays inside the metal over a characteristic skin depth, δ, at which the field falls to 1/e of its surface value. The skin depth depends on frequency f, the material magnetic permeability μ, and electrical conductivity σ. For typical mobile signals around 2.5 GHz, inserting aluminum parameters into the skin depth expression yields a skin depth of roughly 1.6 μm. Household aluminum foil is about 50 μm thick, so it is on the order of 30 skin depths thick, which reduces the transmitted wave amplitude by a factor of roughly e?30, i.e., effectively negligible.
In addition, before any wave penetrates the foil, a large fraction is reflected at the surface. For normal incidence, the reflectivity of aluminum in the microwave and RF bands is extremely high, leaving only a tiny fraction that actually enters the metal. Combining reflection and absorption, wrapping a phone with household aluminum foil can reduce received electromagnetic power by on the order of 17–18 decades (orders of magnitude). Mobile phone receivers operate over roughly 7 decades of signal power, so in theory aluminum foil wrapping can block mobile signals. This is also straightforward to verify experimentally at home.
Note: this explanation is intended to clarify the shielding mechanism rather than raise concerns about handset emissions.
Q4: Why do people without myopia perceive objects as smaller when wearing myopic (concave) glasses?
Myopic glasses are concave (negative lenses) and have a diverging effect on light rays. A concave lens produces a virtual, reduced image on the same side of the lens as the object. That is why, for someone without myopia, viewing objects through myopic glasses produces a smaller virtual image.
Conversely, corrective lenses for farsightedness (convex lenses) act as magnifiers for close objects, producing an enlarged upright virtual image when the object is within the lens focal length; for distant objects, a convex lens may produce an inverted reduced real image depending on geometry.
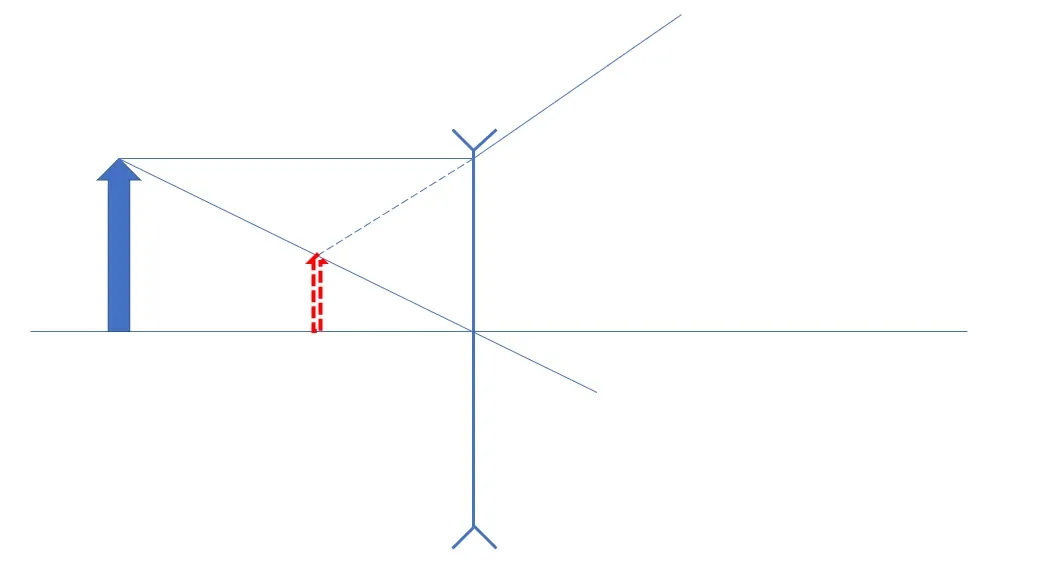
Q5: Why do light sources appear as concentric rings when glasses fog up?
The effect is analogous to rainbow formation. When many small water droplets or a thin film of condensation are present on the lens, light from a source undergoes refractions and internal reflections within individual droplets, producing circular bands around the source. The angular separation of colors arises because different wavelengths refract by different amounts in water, so the scattered light shows color dispersion similar to a rainbow. For artificial light sources, the rings may reflect the source's spectral composition.
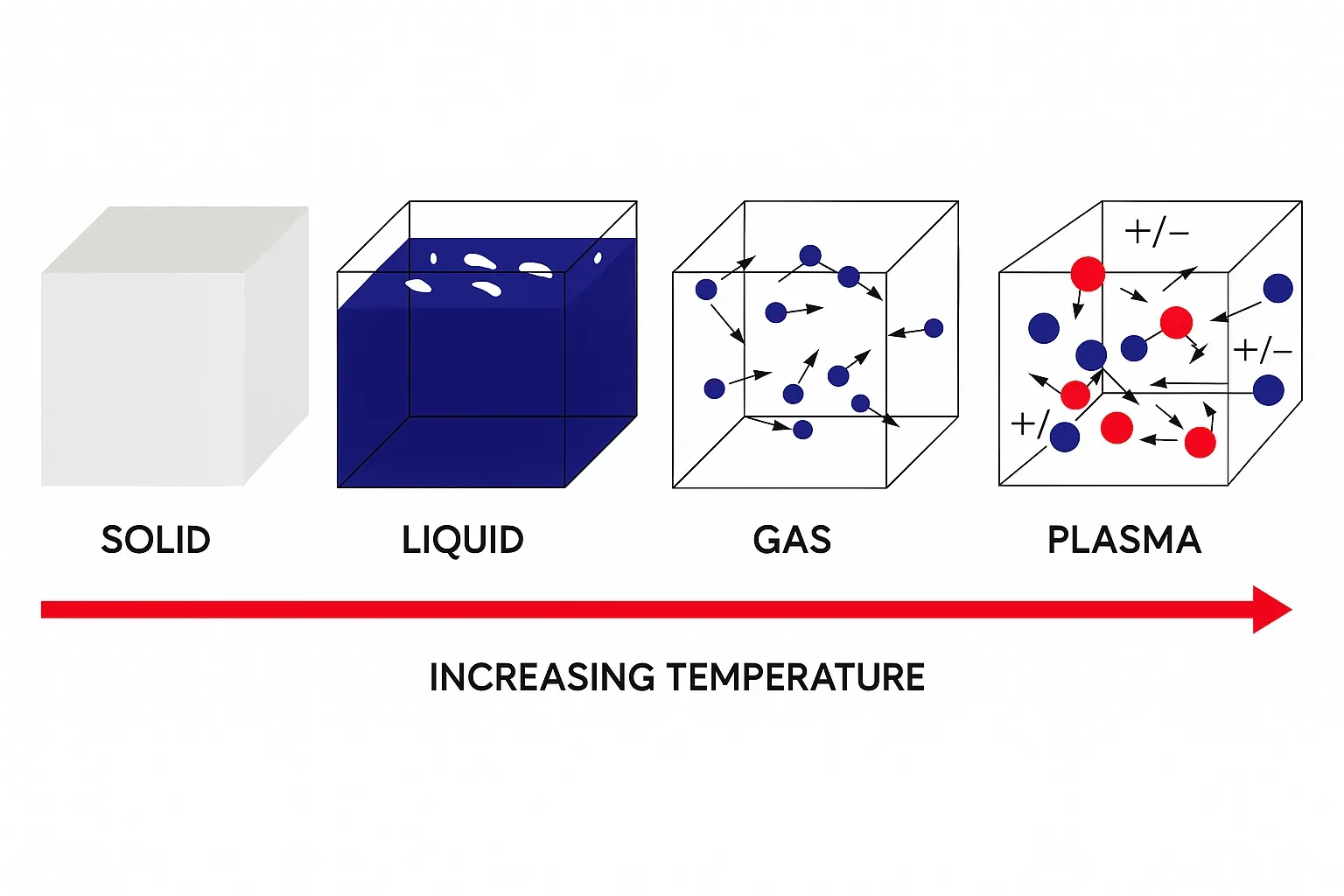
Q6: What state of matter is fire?
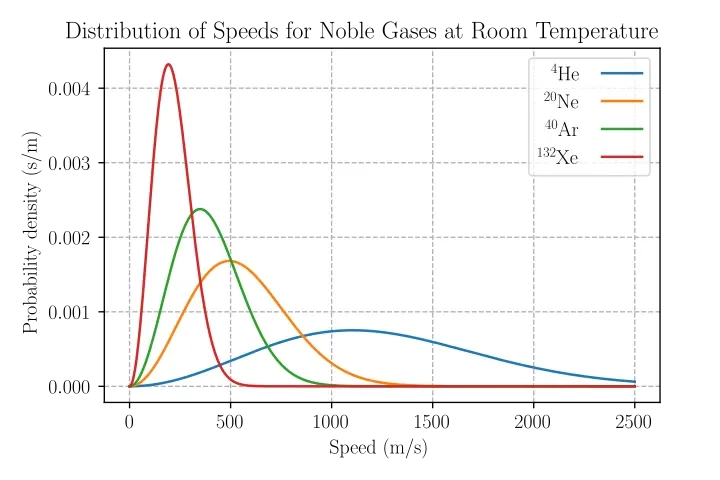
Intuitively, fire is neither solid nor liquid. The most likely classification is that flame is a gas. Some sources label flame as plasma, which can be confusing because gases and plasmas share properties such as variable shape and volume. However, gases and plasmas differ fundamentally: composition (gases consist mainly of neutral molecules; plasma contains a significant fraction of charged particles), electrical conductivity (plasmas are highly conductive), particle velocity distributions, and long-range collective electromagnetic interactions that are present in plasmas but not in neutral gases.
To convert a gas into a plasma typically requires very high temperatures or strong electric fields. For air at atmospheric pressure, temperatures on the order of 14,000 °C would be needed to produce substantial ionization. Common flame temperatures are much lower: charcoal 750–1200 °C; methane 900–1500 °C; propane torch 1200–1700 °C; candle flame mainly up to 1100 °C with hotspots possibly 1300–1400 °C; magnesium 1900–2300 °C; acetylene torch up to 2300 °C; oxyacetylene up to 3300 °C; common lighter flame 280–500 °C. Therefore, most ordinary flames are heated gases where chemical reactions between gas-phase species and fuel release heat; light emission depends on composition and temperature.
That said, very high-temperature flames can contain small amounts of low-degree ionized plasma, but in typical combustion flames the gas-phase description is appropriate.
References
[1] Flame - Wikipedia
[2] Is fire a plasma?
[3] Plasma - Wikipedia
[4] Plasma - Encyclopaedia entry
[5] Maxwell–Boltzmann distribution - Wikipedia
Q7: What would happen if the atmosphere were pure oxygen without other gases?
If the atmosphere were suddenly replaced by pure oxygen at the same volume, the consequences would be severe. Oxygen has a different molecular weight than air, which would change atmospheric pressure and could cause pulmonary distress. Higher oxygen concentration increases metabolic rates and can accelerate aging; prolonged exposure to high oxygen partial pressures can cause oxygen toxicity. Plants would no longer have CO2 for photosynthesis, disrupting primary production and collapsing food chains. The absence of atmospheric nitrogen would break nitrogen cycles essential to life. Anaerobic organisms would be negatively affected. Additionally, higher oxygen concentration greatly increases flammability, making wildfires far more likely.
In Earth's history there have been episodes of elevated atmospheric oxygen. For example, during the Carboniferous period oxygen concentration reached around 35%, which likely supported larger arthropods. However, high oxygen levels also increase the risk of widespread fires.
Overall, replacing the air with pure oxygen would be ecologically catastrophic. Small increases in oxygen fraction may be tolerable, but even modest changes would require careful assessment of ecosystem impacts.
References
[1] Studies on Proterozoic oxygenation events and climate-ecological effects
[2] Geological history of atmospheric oxygen and biodiversity
Q8: If two identical universes were simulated from identical initial conditions, would they evolve identically?
Not necessarily. Quantum mechanics introduces inherent randomness, so evolution is described probabilistically rather than deterministically. Experimental tests of Bell inequalities indicate that local hidden-variable theories cannot reproduce quantum predictions, supporting genuine randomness in quantum phenomena.
Even within classical mechanics, determinism can fail in certain mathematical constructions. The Norton dome example shows that for a particular non-Lipschitz surface, a particle placed at rest on top can remain at rest for an indefinite time and then spontaneously start moving at an arbitrary time. The underlying second-order differential equations admit non-unique solutions because they violate the Lipschitz condition required for uniqueness. This demonstrates that identical initial data need not always produce a unique classical evolution in all mathematical models.