Introduction
Insulin pumps represent a critical advancement in diabetes management, delivering precise insulin doses continuously to patients. The PCB assembly process for these devices demands exceptional reliability due to their life-sustaining function and compact form factor. Optimizing insulin pump PCB assembly ensures minimal failure rates, longevity in body-worn environments, and compliance with rigorous quality benchmarks. Factory teams focus on surface mount technology (SMT) to handle dense component layouts while mitigating risks like solder defects or contamination. This article explores proven strategies to streamline the process, from stencil printing to final inspection, drawing on established manufacturing insights.

Why Insulin Pump PCB Assembly Matters
Insulin pump PCB assembly involves integrating microcontrollers, sensors, batteries, and actuators into small, lightweight boards that must withstand moisture, movement, and thermal cycles. Unlike consumer electronics, these assemblies operate in direct contact with the body, requiring zero-tolerance for defects that could lead to dosing errors or device failure. Manufacturers prioritize high-density interconnects and fine-pitch components to enable miniaturization without compromising signal integrity. Effective optimization reduces rework, accelerates time-to-market, and supports scalability for high-volume production. In factory settings, this process directly impacts patient safety and regulatory approval pathways.
The stakes elevate the need for standardized workmanship, particularly under IPC Class 3 guidelines, which define high-reliability criteria for assemblies where downtime is unacceptable. Engineers and procurement teams benefit from understanding these nuances to select capable partners.
Core Technical Principles in SMT Assembly for Insulin Pumps
SMT assembly for insulin pumps begins with solder paste printing, where precise stencil alignment deposits uniform paste volumes on pads. Factories use laser-cut stencils with apertures tailored to component sizes, often 0.3mm pitch or finer, to prevent bridging during reflow. Component placement follows, employing high-speed machines with vision systems to achieve sub-50 micron accuracy, essential for handling 01005 resistors or QFN packages common in pump designs. This stage minimizes offsets that could cause open joints or tombstoning.
Reflow soldering medical PCB solidifies these connections through controlled heating zones that melt paste without damaging sensitive components. Profiles typically feature gradual ramps to preheat, peak temperatures for reflow, and rapid cooling to form robust intermetallics. Nitrogen atmospheres in ovens reduce oxidation, enhancing joint reliability in humid or biocompatible environments. Factories monitor zone temperatures and conveyor speeds to maintain process windows, avoiding defects like head-in-pillow that plague dense boards.
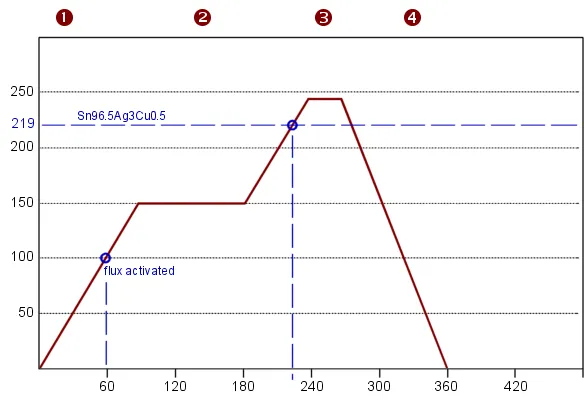
Post-reflow, boards undergo cleaning to remove flux residues, preventing dendritic growth over time. Ultrasonic or vapor degreasing methods ensure ionic contamination levels stay below thresholds, critical for long-term functionality in insulin delivery systems.
Advanced Inspection Techniques: AOI and X-Ray for Insulin Pumps
Automated optical inspection (AOI) insulin pump boards at multiple stages, starting post-paste printing to verify volume and alignment. Multi-angle cameras and algorithms detect insufficient paste, smearing, or stencil wear, enabling immediate corrections before placement. After reflow, AOI scrutinizes top-side joints for fillet shape, wetting, and polarity errors, achieving detection rates that manual checks cannot match. Factories integrate inline AOI to provide real-time feedback, boosting first-pass yields.
For hidden features like BGAs or bottom-side vias, X-ray inspection PCB reveals voids, cracks, or misalignments invisible to optics. Laminar flow X-ray systems generate 2D or 3D images, quantifying solder ball stand-off and fill percentages. This non-destructive method is indispensable for insulin pump assemblies with stacked dies or embedded passives, where joint integrity affects vibration resistance. Combining AOI with X-ray forms a comprehensive quality gate, aligning with J-STD-001 requirements for soldering workmanship.

Best Practices for Optimizing the Assembly Process
Factories optimize insulin pump PCB assembly by standardizing stencil design, opting for electroformed nickel for durability and electro-polished surfaces for clean release. Solder paste selection emphasizes low-voiding formulations with fine particle sizes, suited to lead-free processes. Implementing solder paste inspection (SPI) post-printing maintains volume consistency within 10-25% tolerances, reducing reflow variability.
Placement optimization involves fiducial marks on boards for machine calibration and feeder setup audits to prevent wrong-component errors. Reflow ovens require periodic profiling with thermocouples attached to test boards mimicking pump layouts. Nitrogen purging at 99.99% purity minimizes dross and improves cosmetics on exposed pads.
Cleaning protocols extend to plasma treatment for activated flux removal, followed by ion chromatography to verify residue levels. Conformal coating application protects against moisture ingress, with selective masking for test points. Throughout, traceability via barcode scanning links every board to process data, facilitating root-cause analysis.
Factory Troubleshooting Insights for Common Challenges
Warpage from reflow poses a frequent issue in thin, multilayer boards for insulin pumps, exacerbated by asymmetric copper distribution. Factories counter this with low-CTE substrates and fixture support during heating, measuring bow and twist per IPC-A-600 criteria. Component shifting during cooling affects fine-pitch QFPs; slowing ramp-down rates stabilizes placement.
Contamination from handling introduces ionic risks, addressed by ESD-safe workflows and glove protocols. If voids exceed limits in BGAs, factories adjust paste rheology or preheat dwells. Yield dips prompt DOE runs isolating variables like humidity control in storage. These factory-driven fixes, rooted in data logging, ensure consistent output.
Conclusion
Optimizing insulin pump PCB assembly hinges on precision across SMT processes, from paste deposition to X-ray validation, yielding reliable devices that enhance patient outcomes. Factory adherence to standards like ISO 13485 fortifies quality management, minimizing risks in high-stakes applications. By integrating inline inspections and process controls, manufacturers achieve superior yields and compliance. Procurement teams should prioritize partners with proven Class 3 expertise to navigate miniaturization demands effectively.
FAQs
Q1: What role does SMT assembly play in insulin pump PCB assembly?
A1: SMT assembly enables dense packing of small components like sensors and ICs, vital for compact insulin pumps. Factories optimize pick-and-place accuracy and reflow profiles to form reliable joints under IPC Class 3. This approach supports miniaturization while maintaining functionality in body-worn conditions.
Q2: How does reflow soldering impact medical PCB quality?
A2: Reflow soldering medical PCB requires precise thermal profiles to avoid defects like voids or delamination. Nitrogen use and post-reflow AOI ensure joint integrity per J-STD-001. Factories profile ovens regularly for consistency, enhancing long-term reliability in insulin delivery systems.
Q3: Why is automated optical inspection (AOI) essential for insulin pump boards?
A3: Automated optical inspection (AOI) insulin pump assemblies detects placement errors and solder anomalies early, reducing rework. Inline deployment post-paste and reflow boosts yields in high-volume runs. It complements X-ray for comprehensive coverage, aligning with factory quality gates.
Q4: When should X-ray inspection be used in insulin pump PCB assembly?
A4: X-ray inspection PCB verifies hidden BGA joints and vias undetectable by AOI, critical for multilayer insulin pump designs. Factories apply it post-reflow to quantify voids and alignment. This step ensures vibration resistance and compliance in medical applications.
References
IPC-A-610H — Acceptability of Electronic Assemblies. IPC, 2019
J-STD-001H — Requirements for Soldered Electrical and Electronic Assemblies. IPC, 2018
IPC-A-600K — Acceptability of Printed Boards. IPC, 2020
ISO 13485:2016 — Medical devices — Quality management systems — Requirements for regulatory purposes. ISO, 2016