Introduction
Insulin pumps deliver life-saving medication through precise control systems powered by printed circuit boards. These devices operate continuously in close proximity to the human body, facing unique challenges like skin contact, temperature variations, and mechanical flexing. Proper insulin pump PCB material selection directly impacts patient safety and device longevity. Electrical engineers must prioritize materials that offer biocompatibility, thermal stability, and signal integrity. High-quality materials prevent failures that could lead to dosing errors or device malfunction. This article provides structured guidance on evaluating PCB substrates for these critical applications.
Why PCB Material Selection Matters for Insulin Pumps
Insulin pumps function as wearable medical devices, requiring PCBs that maintain performance under body temperature, humidity, and motion. Standard materials may degrade over time, leading to warpage, delamination, or signal loss. Biocompatible PCB materials ensure no adverse reactions during skin contact or incidental fluid exposure. High TG PCB options resist deformation during manufacturing and operation, enhancing reliability. Thermal conductivity PCB properties help manage heat from microprocessors and batteries. Ultimately, material choices align with the need for uninterrupted service in a compact form factor.
The stakes are high, as any PCB failure can compromise insulin delivery accuracy. Engineers evaluate materials based on long-term stability in physiological environments. Moisture resistance prevents corrosion from sweat or cleaning agents. Dimensional stability supports fine-pitch components common in pump electronics. Selecting suboptimal materials risks regulatory non-compliance and field returns. Thoughtful selection extends device lifespan beyond five years.
Key Material Properties for Insulin Pump PCBs
Biocompatibility stands as a primary requirement for materials in insulin pumps. Substrates must exhibit low extractables and no cytotoxicity to avoid skin irritation or systemic effects. Testing per relevant standards confirms suitability for prolonged body contact. Polyimide and similar polymers offer proven performance in this area. Coverlayers and solder masks also require biocompatibility certification. These properties safeguard patient health while enabling compact designs.
High TG PCB materials provide essential thermal resistance. The glass transition temperature marks where the substrate shifts from rigid to compliant, and elevated values prevent softening during lead-free soldering or elevated operating conditions. This reduces risks of via cracking or board warpage. Compared to standard laminates, high TG variants maintain mechanical strength under stress. They suit multilayer boards with dense routing. Reliability improves in thermal cycling scenarios typical of wearable use.
Thermal conductivity PCB characteristics address heat dissipation in power-sensitive circuits. Processors monitoring glucose levels generate localized heat that standard substrates struggle to spread. Materials with enhanced conductivity, such as ceramic-filled composites, equalize temperatures across the board. This prevents hotspots that degrade components or adhesives. Vias and embedded planes complement material choices for optimal management. Balanced thermal design supports battery efficiency and longevity.
Low coefficient of thermal expansion matches copper to minimize stress in assemblies. Moisture absorption rates influence insulation resistance over time. Dielectric constants affect RF performance for wireless communication modules. Engineers specify based on frequency needs, often favoring low-loss dielectrics. Mechanical flexibility aids rigid-flex hybrids common in pumps. Comprehensive property evaluation ensures holistic reliability.
Common PCB Material Options for Medical Applications
Standard FR-4 serves as a baseline for cost-effective rigid boards, but high TG FR-4 upgrades address thermal demands. These materials withstand multiple reflow cycles without compromising integrity. They find use in control sections of insulin pumps where rigidity supports through-hole components. Biocompatible enhancements, like halogen-free formulations, broaden applicability. Availability and process compatibility make them practical starting points. Limitations in high-frequency performance prompt alternatives for RF sections.
Polyimide substrates excel in flexible and rigid-flex configurations. Their inherent flexibility accommodates pump ergonomics and movement. Thermal stability supports high-density interconnects. Biocompatibility suits skin-contact wearables. Low moisture uptake resists delamination in humid conditions. These materials enable thinner profiles for discreet designs.
PTFE-based laminates, akin to Rogers PCB for medical applications, deliver low dielectric loss for Bluetooth and telemetry. Their stability at microwave frequencies ensures accurate data transmission. Chemical inertness enhances biocompatibility. Thermal conductivity PCB variants within this family aid heat spreading. Rigidity suits antenna modules. Cost trade-offs justify selective use.
Liquid crystal polymer offers ultra-low moisture absorption and barrier properties. It performs well in implantable or semi-implantable contexts. RF compatibility supports miniaturized antennas. Biocompatibility testing confirms safety. Thermal management benefits from matched expansion coefficients. These substrates integrate into hybrid stacks.
Ceramic-filled materials provide superior thermal conductivity PCB performance. They excel in power sections dissipating battery heat. Low expansion prevents cracking. Biocompatibility requires encapsulation. High cost limits to critical zones. Hybrid approaches combine with organics for balance.
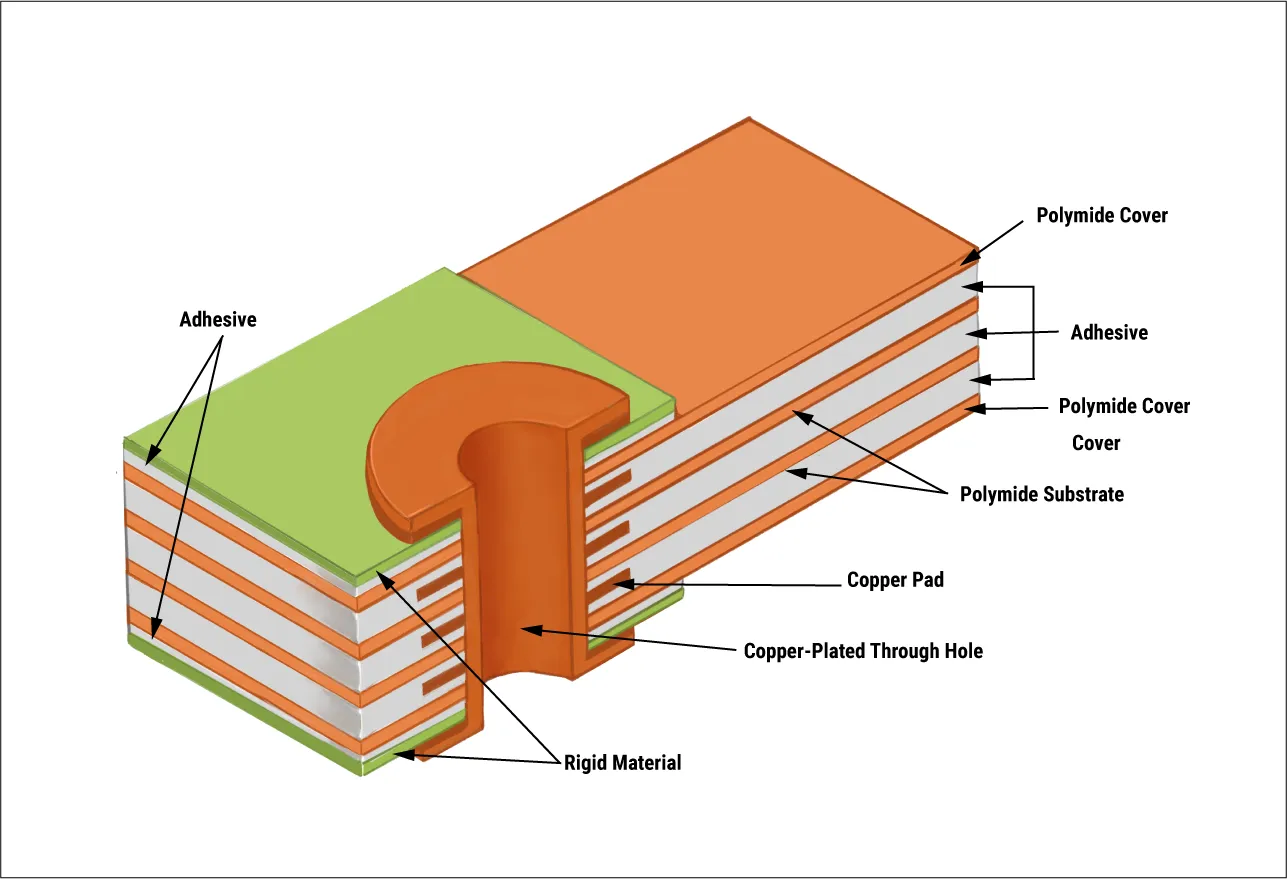
Per IPC-6012, qualification verifies performance across these options.
Standards and Compliance in Material Selection
Industry standards guide insulin pump PCB material selection to ensure repeatability and safety. IPC-6012 outlines qualification for rigid boards, emphasizing thermal shock and electrical testing. Compliance confirms materials endure manufacturing stresses. Class 3 provisions apply to high-reliability medical uses.
Assembly follows IPC-A-610 criteria for acceptability. Visual and dimensional inspections verify material integrity post-process. Solder joint reliability ties to substrate properties. These guidelines prevent field failures.
IEC 60601 addresses electrical safety in medical equipment. Material choices influence insulation and leakage currents. Thermal endurance supports continuous operation ratings.
Testing includes accelerated aging to simulate years of use. Moisture sensitivity per J-STD-020 classifies handling. Engineers document traceability for audits.
Best Practices for Insulin Pump PCB Material Selection
Begin with application mapping to identify thermal, mechanical, and electrical hotspots. Prioritize biocompatible PCB materials for external sections. High TG PCB for reflow-exposed areas. Simulate thermal profiles early.
Collaborate with fabricators on process capabilities. Validate stackups for warpage control. Use thermal vias where conductivity falls short.
Prototype and test per standards. Monitor impedance for RF sections. Encapsulate with parylene for added protection.
Iterate based on failure analysis. Balance cost with performance. Document rationale for regulatory submission.
Troubleshooting Common Material-Related Issues
Warpage arises from CTE mismatch, addressed by high TG selection and symmetric stacks. Delamination links to moisture, mitigated by low-absorption substrates. Signal loss in RF traces demands low-loss dielectrics like those in Rogers PCB for medical applications.
Overheating prompts thermal conductivity PCB enhancements or redesigns. Biocompatibility failures trace to extractables, resolved by certified materials. Field returns often stem from untested flex endurance.
Systematic root cause analysis guides fixes. Accelerated testing predicts issues.
Conclusion
Insulin pump PCB material selection demands a multifaceted approach balancing biocompatibility, thermal stability, and performance. High TG PCB, biocompatible PCB materials, and thermal conductivity PCB options form the foundation. Standards like IPC-6012 ensure compliance. Engineers achieve reliable designs through simulation, testing, and iteration. Prioritizing these factors delivers devices that sustain patient health reliably.
FAQs
Q1: What role does high TG PCB play in insulin pump reliability?
A1: High TG PCB materials resist deformation at elevated temperatures, crucial during soldering and body-worn operation. They maintain dimensional stability, preventing via failures and warpage. This enhances overall assembly robustness per IPC standards. Insulin pump PCB material selection favoring high TG extends service life in thermal cycling environments. Electrical engineers specify them for multilayer boards.
Q2: Why are biocompatible PCB materials essential for insulin pumps?
A2: Biocompatible PCB materials prevent skin irritation or toxicity in wearable applications. They exhibit low leachables and pass cytotoxicity tests. Essential for prolonged contact, these substrates support regulatory approval. Polyimide and LCP variants excel here. Proper selection avoids clinical complications.
Q3: How does thermal conductivity PCB impact insulin pump design?
A3: Thermal conductivity PCB properties dissipate heat from ICs and batteries efficiently. This prevents hotspots degrading performance or lifespan. Ceramic-filled options aid compact layouts. Combined with vias, they optimize efficiency. Critical for low-power, continuous operation.
Q4: When to consider Rogers PCB for medical applications in insulin pumps?
A4: Rogers PCB for medical applications suit RF modules needing low loss and stability. They handle high frequencies for wireless links without attenuation. Biocompatibility and thermal traits align with pump needs. Use selectively for antennas amid cost constraints. Evaluate dielectric constants first.
References
IPC-6012E — Qualification and Performance Specification for Rigid Printed Boards. IPC, 2015
IPC-A-610H — Acceptability of Electronic Assemblies. IPC, 2019
IEC 60601-1 — Medical electrical equipment - Part 1: General requirements for basic safety and essential performance. IEC, 2005
J-STD-020E — Moisture/Reflow Sensitivity Classification for Nonhermetic Surface Mount Devices. JEDEC, 2014