Introduction
Printed circuit boards form the foundational backbone of medical devices, integrating complex electronics that must perform flawlessly under continuous operation. In the medical sector, where device failure can lead to severe consequences, ensuring PCB reliability demands more than just design excellence; it requires robust manufacturing processes capable of scaling production without compromising quality. PCB manufacturing capacity refers to a facility's ability to produce boards at required volumes while upholding stringent quality controls, directly impacting the delivery of quality medical devices. Engineers evaluating suppliers must prioritize capacity not as mere throughput, but as a guarantee of consistent Medical Devices Manufacturing Quality across batches. This article explores how manufacturing capacity influences reliability in medical applications, drawing on factory-driven insights to guide procurement and production decisions. By understanding these dynamics, electric engineers can better select partners for high-volume runs that support Reliable Medical Devices Volume.

What Is PCB Manufacturing Capacity and Why It Matters for Medical Devices
PCB manufacturing capacity encompasses the infrastructure, equipment, personnel expertise, and process controls that enable a facility to meet production volumes ranging from prototypes to millions of units. It includes factors like panel throughput rates, multilayer lamination capabilities, and surface finish application speeds, all calibrated to maintain precision at scale. For medical devices, capacity becomes critical because these products often transition rapidly from low-volume validation to high Manufacturing Volume Devices demands driven by regulatory approvals and market needs. A facility with inadequate capacity risks rushed processes, leading to defects such as delamination or via failures that undermine device longevity.
The relevance intensifies in medical contexts, where PCBs must withstand sterilization cycles, humidity exposure, and mechanical stresses without degradation. High capacity ensures statistical process control can be applied effectively, yielding defect rates below acceptable thresholds for life-critical applications. Without sufficient capacity, quality drifts occur due to overburdened equipment or untrained shifts, directly eroding Medical Devices Manufacturing Quality. Electric engineers recognize that true capacity aligns production scalability with reliability metrics, preventing costly recalls and ensuring patient safety.
Key Technical Principles Linking Capacity to Reliability
At the core of PCB reliability lies process stability, which high-capacity manufacturing reinforces through automated handling and real-time monitoring. In multilayer boards common to medical imaging or monitoring devices, precise alignment during lamination prevents misalignment that could cause signal integrity issues. Facilities with robust capacity invest in multiple press lines and optical registration systems, allowing consistent pressure and temperature profiles across production runs. This scalability minimizes warpage, a common reliability killer, by distributing thermal loads evenly during curing.
Material traceability and storage capacity further bolster reliability, as medical PCBs demand controlled environments to prevent moisture absorption in laminates. High-volume operations implement climate-controlled warehouses and baking protocols before assembly, ensuring compliance with handling guidelines. Capacity also enables rigorous incoming inspection of copper foils and prepregs, reducing variability in dielectric properties that affect impedance control. Engineers appreciate how these elements scale to support Quality Medical Devices, where even minor inconsistencies amplify failure risks in vivo.
Inspection methodologies scale differently based on capacity; low-volume setups rely on manual checks, while high-capacity lines deploy automated optical inspection and X-ray systems for 100% coverage. These tools detect voids, cracks, and plating anomalies at rates infeasible in smaller operations. Adherence to IPC-6012 standards for qualification and performance ensures that high-volume processes meet Class 3 criteria for high-reliability boards, verifying conductor width tolerances and solder mask registration.

Solder mask and surface finish application exemplify capacity's role in reliability. Electrochemical processes like ENIG demand stable plating baths, which large-capacity facilities maintain through continuous replenishment and filtration systems. Overextended capacity leads to bath depletion, causing black pad issues that compromise solder joint integrity. Robust capacity supports ionic contamination testing post-fabrication, correlating surface cleanliness to long-term reliability under humidity.
Practical Best Practices for High-Volume Medical PCB Production
To achieve Reliable Medical Devices Volume, factories implement design for manufacturability reviews early, optimizing panel utilization for efficient high-volume runs. Nested layouts maximize throughput while preserving edge clearances for depaneling, reducing stress-induced microcracks. Engineers should specify panel sizes aligned with facility capabilities, ensuring via drilling and routing maintain positional accuracy across arrays. This upfront planning sustains yield rates above 95% in sustained production.
Process validation through capability studies quantifies manufacturing stability, using control charts to monitor critical parameters like etch factor and hole wall roughness. High-capacity operations conduct these studies per shift, adjusting etchant chemistry to counteract cumulative drift. Integrating flying probe testing for electrical opens/shorts scales verification without fixtures, ideal for variant-rich medical designs. Such practices uphold consistent Medical Devices Manufacturing Quality, minimizing field failures.
Cleanroom integration for final bare board inspection prevents particulate contamination, a silent reliability threat in implantable devices. Capacity allows dedicated Class 1000 environments with laminar flow, where operators perform visual and dimensional checks against IPC-A-610 acceptability criteria. Post-inspection baking removes residual moisture, preparing boards for assembly. Electric engineers benefit from suppliers documenting these controls, providing traceability for audits.
Supplier qualification emphasizes quality management systems certified to ISO 13485, which mandates risk-based controls tailored to medical applications. This standard drives capacity planning with validated processes and supplier controls for raw materials. Facilities demonstrate capacity through documented change control, preventing unvalidated shifts during volume ramps. By prioritizing these, procurement teams secure Quality Medical Devices scalable to market demands.
Challenges in Scaling Capacity and Proven Solutions
One persistent challenge in high-volume medical PCB production is maintaining drill accuracy as bit wear accelerates with throughput. Advanced facilities rotate diamond-coated bits and employ adaptive CNC programming to compensate for runout, preserving aspect ratios up to 10:1. Statistical analysis of hole locations flags tooling needs, averting misalignment in HDI structures critical for compact diagnostics.
Yield dilution from mixed lots poses another hurdle; capacity mitigates this via dedicated lines for Class 3 medical runs, isolating processes from consumer volumes. Segregated plating and etch lines prevent cross-contamination, ensuring consistent copper thickness. Engineers monitor CpK values for plating uniformity, targeting above 1.67 for reliability margins.
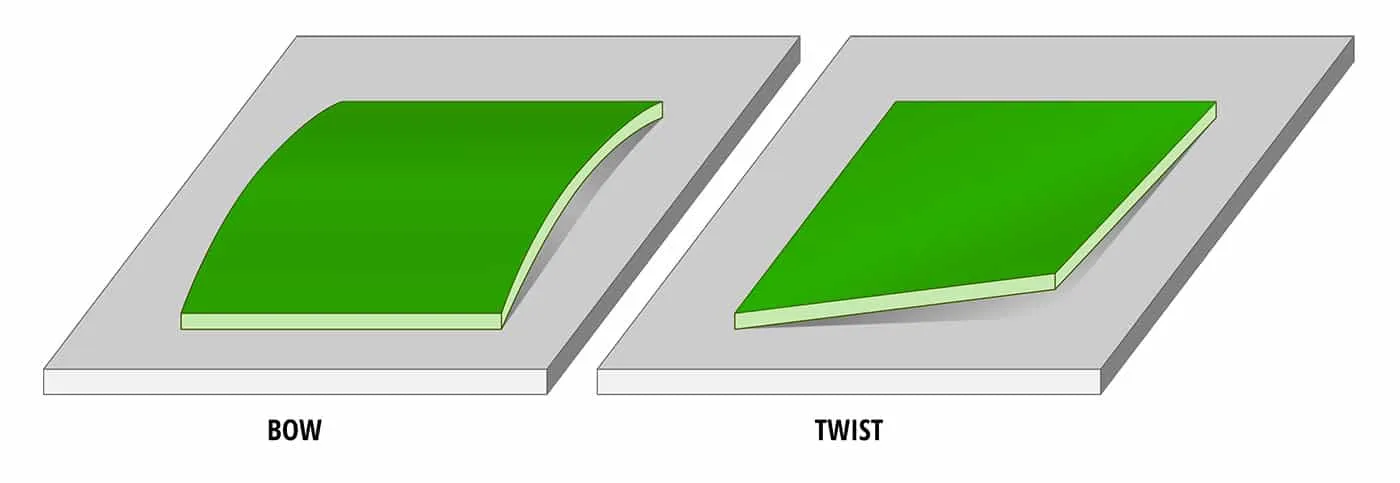
Thermal management during high-volume lamination risks uneven curing, leading to bow and twist. Solutions include zoned presses with independent platens and fixtureless handling for thin cores. Capacity enables post-lam bake cycles to relieve stresses, followed by warpage measurement to confirm flatness within 0.75% limits.
Supply chain volatility affects capacity; proactive sourcing of certified laminates buffers against shortages. Long-lead HDI builds demand forecast sharing, allowing facilities to reserve sequential lamination slots. These strategies ensure Manufacturing Volume Devices without expedited premiums that strain quality.
Conclusion
PCB manufacturing capacity stands as a pivotal determinant of medical device reliability, bridging scalable production with unwavering quality standards. Electric engineers must evaluate facilities holistically, focusing on process controls, inspection scalability, and adherence to benchmarks like IPC and ISO guidelines. By selecting partners with proven high-volume capabilities, teams deliver Quality Medical Devices that perform reliably in clinical settings. Prioritizing capacity fosters innovation in diagnostics and therapeutics, ultimately safeguarding patient outcomes through robust electronics.
FAQs
Q1: What role does PCB manufacturing capacity play in achieving Reliable Medical Devices Volume?
A1: High manufacturing capacity ensures processes remain stable during volume ramps, applying statistical controls to maintain low defect rates. Facilities with ample throughput implement automated inspections and material traceability, preventing quality degradation seen in overburdened lines. This scalability supports market demands for medical devices without compromising reliability, aligning with Class 3 standards for critical applications. Engineers gain confidence in suppliers demonstrating consistent yields at scale.
Q2: How does manufacturing capacity impact Medical Devices Manufacturing Quality?
A2: Capacity enables dedicated cleanrooms, real-time monitoring, and validated tooling that uphold precision across batches. Overextended operations risk process drift, but robust capacity sustains parameters like via fill and surface finish integrity. Integration of AOI and flying probe testing scales quality verification, reducing escapes to implantable devices. Factory insights emphasize capacity as the foundation for repeatable Medical Devices Manufacturing Quality.
Q3: Why is high manufacturing capacity essential for Quality Medical Devices?
A3: Quality Medical Devices demand PCBs free from warpage, contamination, and plating defects, which high capacity safeguards through zoned processing and 100% testing. It allows risk-based qualification per ISO standards, ensuring traceability from laminate to finished board. Engineers prioritize capacity to avoid low-volume biases that fail under field stresses like sterilization. Ultimately, it translates to dependable performance in patient-critical electronics.
Q4: Can PCB manufacturing capacity affect the reliability of high-volume devices?
A4: Yes, capacity directly influences reliability by enabling consistent thermal profiles and inspection coverage at scale. Facilities with excess capacity conduct capability studies, optimizing etch and laminate steps for minimal variability. This prevents issues like microcracks in multilayer stacks, vital for Manufacturing Volume Devices. Proven capacity correlates with extended MTBF in medical applications.
References
IPC-6012E — Qualification and Performance Specification for Rigid Printed Boards. IPC, 2017
IPC-A-610H — Acceptability of Electronic Assemblies. IPC, 2019
ISO 13485:2016 — Medical devices — Quality management systems — Requirements for regulatory purposes. ISO, 2016






