Introduction
In the medical device industry, printed circuit boards (PCBs) serve as critical components that must prioritize patient safety and device reliability. UL certification plays a pivotal role in verifying that medical PCBs meet rigorous safety benchmarks, reducing risks such as electrical shock, fire hazards, and mechanical failure. Engineers designing or procuring UL certified medical PCBs ensure compliance with medical PCB safety standards, which directly impacts regulatory approvals and market access. This certification process evaluates materials, construction, and performance under real-world conditions, providing assurance for applications in diagnostics, monitoring, and therapeutic equipment. As devices become more complex with higher power densities and miniaturization, understanding UL compliance for medical devices becomes essential for electric engineers focused on long-term performance.
What Is UL Certification and Why It Matters for Medical PCBs
UL certification represents an independent evaluation confirming that a PCB adheres to established safety criteria for use in end products. For medical PCBs, this certification is particularly vital because failures can lead to life-threatening consequences, unlike consumer electronics. A UL certified medical PCB undergoes testing for flammability, dielectric breakdown, and spacing to prevent arcing, ensuring it supports the overall device safety profile. Manufacturers apply the UL mark only after factory inspections and ongoing surveillance, fostering trust among regulatory bodies and end-users. In high-stakes environments like hospitals, UL compliance for medical devices minimizes liability while streamlining FDA or equivalent submissions. Engineers benefit from this by selecting boards that align with proven risk mitigation strategies.

Without UL certification, medical devices risk rejection during audits, delaying time-to-market and increasing redesign costs. The process also encourages the use of qualified materials resistant to environmental stressors common in medical settings, such as humidity and sterilization cycles. Ultimately, it bridges design intent with manufacturing reality, enabling engineers to focus on innovation rather than basic safety validation.
Key Safety Requirements in UL Certification for Medical PCBs
UL certification for medical PCBs emphasizes electrical insulation integrity, where minimum creepage and clearance distances prevent unintended current paths. Tests simulate fault conditions, like overvoltage, to confirm the board withstands stresses without propagating hazards to patients or operators. Material selection focuses on low flammability ratings, ensuring self-extinguishing properties during abnormal thermal events. Factory-driven processes, including controlled lamination and plating, maintain uniformity critical for high-reliability applications. Compliance involves detailed documentation of laminate Tg, CTE, and Z-axis expansion to avoid delamination under thermal cycling.
These requirements align with broader medical PCB safety standards, such as those outlined in IEC 60601-1, which specifies general safety for medical electrical equipment. Engineers must verify that PCB layouts incorporate adequate isolation barriers between patient-applied and operator-accessible parts. Solder mask and conformal coatings receive scrutiny for pinhole defects that could compromise insulation resistance over time.

Mechanical robustness testing includes bend, twist, and vibration assessments to simulate handling and transport stresses. Dimensional stability prevents warpage that could misalign components or connectors in assembled devices. UL evaluates production controls, like etch factor control and via plating thickness, to ensure consistent performance across batches.
Technical Mechanisms Behind UL Safety Testing
The core of UL certification lies in standardized test sequences that replicate operational and fault scenarios. Dielectric voltage withstand tests apply high potentials across traces and layers, measuring leakage current to validate insulation strength. Flammability trials expose board sections to open flames, assessing afterglow and drip characteristics for rapid quenching. Thermal shock cycling between extremes, such as minus 65 degrees C and 150 degrees C, checks for cracks in vias or pads that might evolve into opens or shorts.
Production lot qualification involves sectioning boards for metallographic analysis, revealing interlayer adhesion and copper ductility. Engineers analyze these results to refine stackups, balancing signal integrity with safety margins. Humidity bias testing combines elevated temperature, moisture, and voltage to accelerate corrosion mechanisms at conductor edges.
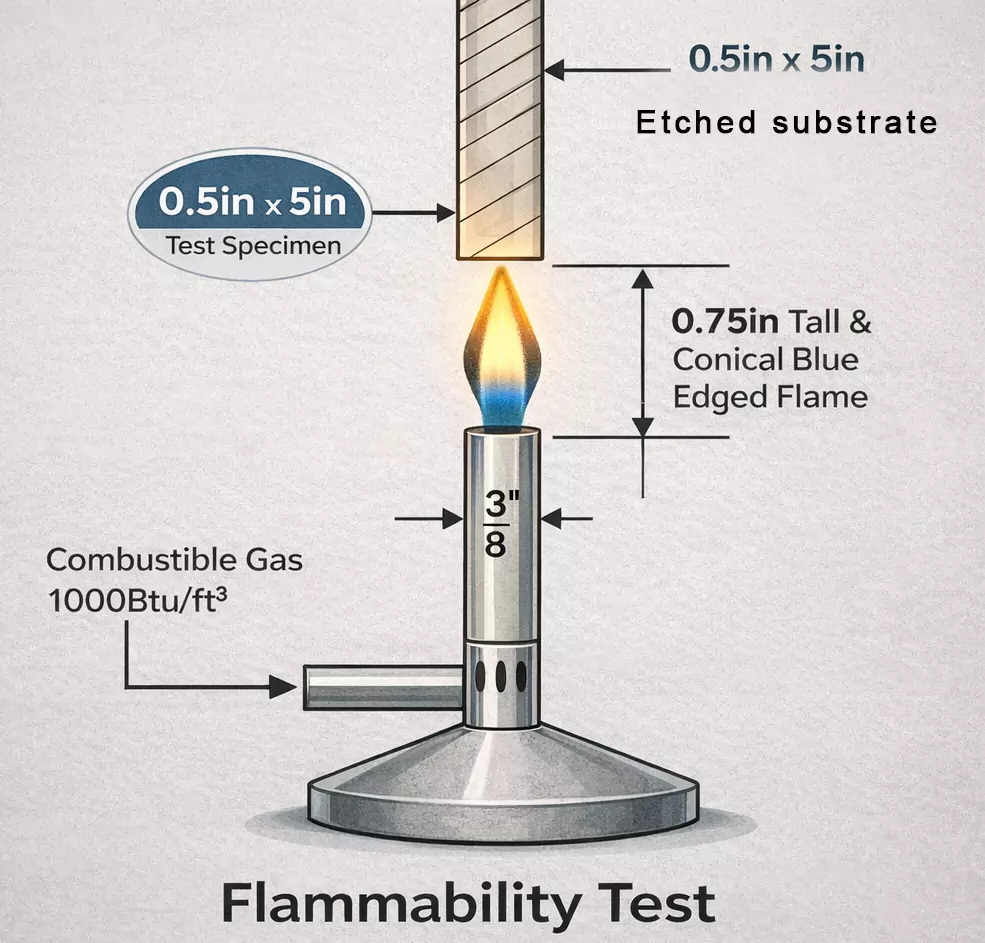
Compliance with IPC-6012F ensures qualification criteria for rigid boards used in demanding sectors, including medical, by defining performance classes like Class 3 for uninterrupted service. This standard guides acceptance of conductor width variations and hole wall quality, directly supporting UL outcomes. Integration of these mechanisms allows PCBs to endure sterilization processes without degrading safety features.
Best Practices for Achieving UL Compliance in Medical PCB Manufacturing
Start with material datasheets specifying UL-recognized grades, focusing on FR-4 variants with high CTI for pollution degree 2 environments. Design rules enforce minimum spacings scaled by working voltage, often exceeding general IPC guidelines for medical conservatism. Prototype builds undergo pre-certification audits to identify gaps early, saving iteration costs.
Implement traceability from raw laminate to finished panel via serialized barcodes, facilitating UL factory follow-up inspections. Process controls include automated optical inspection for solder mask registration and X-ray for barrel fill in high-aspect vias. Cleanliness verification post-fabrication uses ion chromatography to quantify residues that could lower surface insulation resistance.
Adopt ISO 13485 quality management systems to structure documentation and risk assessments, aligning manufacturing with regulatory expectations. Collaborate with certified fabricators experienced in medical volumes, ensuring consistent application of controlled impedance where RF signals demand it. Post-certification, maintain records for recertification cycles, adapting to design evolutions.
Troubleshooting Common Challenges in UL Medical PCB Certification
Engineers often encounter failures in dielectric testing due to inconsistent plating thickness, resolved by optimizing electroplating bath chemistry and current density profiles. Warpage exceeding 0.75% triggers rejection, mitigated through symmetric stackups and low-stress laminates matched for CTE. Flammability issues arise from resin-rich areas; uniform glass cloth distribution during layup prevents this.
If clearance violations appear post-etch, revise panelization to minimize nested features that distort etch uniformity. For assembled boards pursuing UL 60601-1 alignment, verify component spacing complements PCB features. Regular cross-section analysis during qualification lots catches voids early, allowing recipe tweaks before full production.
Conclusion
UL certification stands as a cornerstone for medical PCBs, guaranteeing they meet medical PCB safety standards through exhaustive safety validations. Electric engineers leverage this to deliver reliable UL compliance medical devices, integrating standards like IEC 60601-1 and IPC-6012F into workflows. Factory insights emphasize proactive controls, from material selection to final inspections, ensuring batch-to-batch consistency. By prioritizing these practices, teams reduce risks, accelerate approvals, and enhance device longevity in clinical use. Ultimately, a UL certified medical PCB not only satisfies regulators but elevates overall system integrity.
FAQs
Q1: What makes a UL certified medical PCB essential for compliance with medical PCB safety standards?
A1: A UL certified medical PCB undergoes rigorous testing for electrical, thermal, and mechanical safety, aligning with requirements like isolation barriers and flammability resistance. This certification verifies the board's suitability as a component in patient-contact devices, preventing hazards such as shock or fire. Engineers rely on it to demonstrate risk management per established protocols, streamlining regulatory reviews. Factory processes ensure traceability, supporting sustained performance.
Q2: How does UL compliance for medical devices integrate with IPC standards?
A2: UL compliance for medical devices builds on IPC-6012F Class 3 guidelines, which define qualification for high-reliability rigid boards. This includes tolerances for conductor geometry and hole quality critical for medical applications. Engineers use these to design layouts that pass UL evaluations without rework. The synergy reduces failure modes, enhancing overall device safety.
Q3: What role does IEC 60601-1 play in UL certification for medical PCBs?
A3: IEC 60601-1 establishes baseline safety for medical electrical equipment, influencing UL tests for creepage, clearance, and leakage current on PCBs. UL certification confirms the board supports these requirements, protecting against patient-applied part risks. Electric engineers incorporate its principles during layout to avoid certification hurdles. Compliance fosters interoperability in complex systems.
Q4: Why prioritize ISO 13485 in manufacturing UL certified medical PCBs?
A4: ISO 13485 provides a framework for quality systems tailored to medical devices, ensuring consistent processes for UL certified medical PCB production. It mandates risk-based controls for fabrication steps like lamination and etching. Engineers benefit from auditable records that bolster UL surveillance. This standard minimizes variability, upholding safety standards long-term.
References
IEC 60601-1 — Medical electrical equipment - Part 1: General requirements for basic safety and essential performance. IEC, 2005
IPC-6012F — Qualification and Performance Specification for Rigid Printed Boards. IPC, 2023
ISO 13485:2016 — Medical devices - Quality management systems - Requirements for regulatory purposes. ISO, 2016







